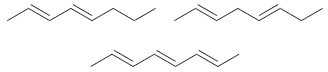
Rank the following compounds in order of increasing λ max :
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)

Answered By

Rinki Devi
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and helped them achieve great subject knowledge.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Rank the following compounds in order of increasingacidity: (a) CH3CH2CO2H (b) CH3CH2OH (c) (CH3CH2)2NH (d) CH3COCH3 (e) (f) CCI3CO2H CCH-CCH3
-
Rank the following compounds in order of increasing basicity. CH OH NH
-
1. Rank the following compounds in order of increasing acidity. 2. Indicate which compounds would be more than 99% deprotonated by a solution of sodium ethoxide in ethanol. () (b) () (d) () (f)...
-
In Pissarides's view of labor markets (job matching model), the focus is on finding a good ________. a. level of TFP (A) b. supply of money (M). c. match d. Wage
-
Given the vertical and horizontal normal stresses of Problem 11.12 Find the maximum values of shear stress on the horizontal and vertical planes to cause failure in a medium dense sand. Assume the...
-
Dr. Randolph, a cash basis taxpayer, knows that he will be in a lower marginal tax bracket next year. To take advantage of the expected decrease in his tax rate, Dr. Randolph instructs his office...
-
A six-cylinder, 4-stroke SI engine delivers \(400 \mathrm{~kW}\) at \(2200 \mathrm{rpm}\). Determine the bore and stroke from the following data : Compression ratio \(=7.6 ;\) stroke to bore ratio...
-
Refer to Figure which shows the computer solution THE MANAGEMENT SCIENTIST SOLUTION FOR THE INVESTMENT ADVISORS PROBLEM a. How much would the return for U.S. Oil have to increase before it would be...
-
II) (4 points) Consider the digital circuit shown in figure below. Write a Boolean equation (function) to compute the circuit's output M in terms of the circuit's inputs (i.e. M = f(x3, x2, x1, x 0,...
-
Papas Pizza Heaven serves take-out pizza from three locations in Columbus, Maryland. Papas considers each pizza delivered (even if the order is for two or more pizzas) as the cost object for the...
-
To an economist, any government program is too big if an analysis of that program finds that MB _______ MC. a. Is greater than b. Is less than c. Is equal to d. Is less than twice as large as e. Is...
-
What are two determinants of species richness? Give an example of each.
-
From the given income statement and additional information of Carlos Co., compute the following: a. Asset turnover for 2013 b. Inventory turnover for 2013 c. Accounts receivable turnover for2013 Net...
-
Gamma Corporation, an S corporation, has a fiscal year ending March 31. It is required to switch to a calendar-year tax year. How many months of income would a calendar- year taxpayer be required to...
-
Several individuals form Lang Corporation on May 1, 2019. The corporation begins acquiring assets on June 1, 2019, and begins business on August 1, 2019. What is the latest date that Lang can file...
-
Alpha Corporation began business at the beginning of the year. On February 15, George sold his stock to Danny. Alpha files for S corporation status on March 10, but George refuses to consent to the...
-
Damion Corporation begins its first tax year on June 23, 2019. What is the latest date that Damion may apply for S corporation status to qualify as of the beginning of operations?
-
Acme Corporation, a calendar year S corporation, has $100,000 of Accumulated Earnings and Profits. Larry, the sole shareholder, has adjusted basis of $80,000 in stock basis with a $20,000 in...
-
Which of the following is responsible for processing a web pages html instructions? A. Client computer b. Web server
-
Cassandra Casey operates the Futuristic Antique Store. She maintains subsidiary ledgers for accounts payable and accounts receivable. She presents you with the following information for October 2019:...
-
Write resonance structures for the followinganions: (a) (b) {c) -, N=CCHCOCH3 CHH CH3CH (e) (d) H OCH3
-
Predict the product(s) of the followingreactions: (b) 1. Nat "OEt Co (a) 2. CH3I eat (c) Br2, PBr3 H20 CH-CH2C (d) " NaOH, H20 12
-
Which, if any, of the following compounds can be prepared by a malonic ester synthesis? Show the alkyl halide you would use in each case. (a) Ethyl pentanoate (b) Ethyl 3-rnethylbutanoate (c) Ethyl...
-
Choose from the following topics to create discussion: Humanity and the Natural Environment Housing and Home Furnishings Privacy and Socializing
-
On December 31, 2020, Pearl Company had $1,101,000 of short-term debt in the for January 21, 2021, the company issued 22,800 shares of its common stock for $42 per brokerage fees and other costs of...
-
During your interview process for the position of program manager for Booker, Tee, and How you are asked to explain the relationship between cost, schedule, and performance? what is your answer to...

Study smarter with the SolutionInn App


