Single bonds generally experience free rotation at room temperature: Nevertheless, the single bond shown below exhibits a
Question:
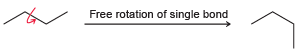
Nevertheless, the €œsingle bond€ shown below exhibits a large barrier to rotation. In other words, the energy of the system is greatly increased if that bond is rotated. Explain the source of this energy barrier. (Hint: Think about the atomic orbitals being used to form the €œconduit.€)
Transcribed Image Text:
Free rotation of single bond
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The single bond mentioned in this pr oblem has some double bond character ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Sigma bonds experience free rotation at room temperature: In contrast, Ï bonds do not experience free rotation. Explain. . cec - `H
-
At room temperature the electrical conductivity and the electron mobility for copper are 6.0 107 (-m)-1 and 0.0030 m2/V-s, respectively. (a) Compute the number of free electrons per cubic meter for...
-
Proton NMR spectroscopy provides evidence for the restricted rotation of a peptide bond. For example, N,Ndimethylformamide exhibits three signals in its proton NMR spectrum at room temperature. Two...
-
Zanny Mouldings has the following estimated costs for the year ahead: Directly used materials $25,100 Direct labor costs $62,300 factory chief salary $37,800 Ad spend 34.000 $ Heating and lighting...
-
Suppose you have an array of N elements containing only two distinct keys, true and false. Give an O(N) algorithm to rearrange the list so that all false elements precede the true elements. You may...
-
Outdoor Furniture Company manufactures outdoor furniture from recycled plastic lumber (HDPE). The following table contains events that occur in the manufacture and sale of tables by Outdoor Furniture...
-
With reference to Exercise 11.16, find a \(90 \%\) confidence interval for \(\alpha\). Data From Exercise 11.16 11.16 Recycling concrete aggregate is an important compo- nent of green engineering....
-
On January 1, 2011, Cam borrows $400,000 from Ven. The five-year term note is a variable-rate one in which the 2011 interest rate is determined to be 8 percent, the LIBOR rate at January 1, 2011, +...
-
TER Inc. had one temporary difference at the end of 2020 that will reverse and cause a taxable amount of 1,100,000 in 2021 and 1,200.000 in 2022. The entitv has a deductible temporary difference of...
-
Suppose you are conducting market research for your favorite soda brand. Sales have been lagging for two quarters, and you are determined to find out why. You decide to host an in-person focus group...
-
The Medicare Trust Fund is necessary because a. Current expenses are greater than current revenues. b. Current expenses are less than current revenues. c. Future expenses will be greater than future...
-
Per capita real economic growth during the Industrial Revolution through the mid-1970s was a. Negative. b. Zero. c. 1 percent. d. 2 percent.
-
Perform the operation. Write answers in standard form. (5 - 11i) (5 + 11i)
-
Is risk-neutrality an appropriate assumption for benefitcost analysis? Why or why not? Does it seem more appropriate for some environmental problems than others? If so, which ones? If you were...
-
Certain environmental laws prohibit EPA from considering the costs of meeting various standards when the levels of the standards are set. Is this a good example of putting first things first or...
-
What is e-business? (Section 25.7) Data from section 25.7 This section gives a very brief summary of some aspects of e-business and shows how management accounting has a role to play in e-business....
-
Explain how the structure of the statement of cash flows represents another subsection of the accounting equation. (Section 3.6) Data from section 3.6 It was shown in Chapter 1 that liquidity is of...
-
The group makes full provision for the future cost of decommissioning oil and natural gas production facilities and related pipelines on a discounted basis on the installation of those facilities....
-
Evaluate the following limits or explain why they do not exist. Check your results by graphing. cos 0 lim_ (tan 0)os cos e 0>T/2
-
A company produces earbuds. The revenue from the sale of x units of these earbuds is R = 8x. The cost to produce x units of earbuds is C = 3x + 1500. In what interval will the company at least break...
-
The amino acid phenylalanine has pKa = 1.83, and tryptophan has pKa = 2.83. Which is the strongeracid? OH H3N H H3N H Tryptophan Phenylalanine (pka = 1.83) (pka = 2.83)
-
Amide ion, H 2 N , is a stronger base then hydroxide ion, HO . Which is the stronger acid, NH 3 or H 2 O? Explain.
-
Will either of the following reactions take place as written, according to the data inTable? (a) HCN + CH3CO2 Na+ Na* "CN CHCO2H Na* "CN CH3CH20- Na* |(b) CH3CH2OH HCN
-
What interventions are the most beneficial to the clients your agency serves? Explain
-
Using the following predicates: square (x) is true if x is a square (otherwise it is false) star (x) is true if x is a star (otherwise it is false) circ(x) is true if x is a circle (otherwise it is...
-
Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any common stock during the year. A total of...

Study smarter with the SolutionInn App


