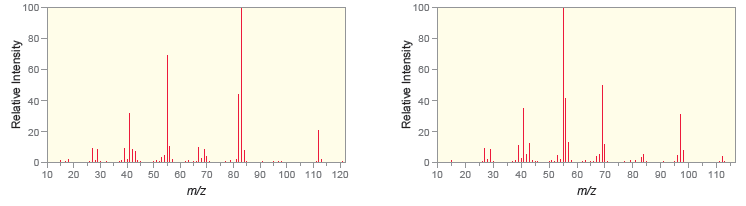
The following are mass spectra for the constitutional isomers ethylcyclohexane and 1, 1-dimethylcyclohexane. Based on likely fragmentation
Question:
Transcribed Image Text:
100 100 80 - 80- 60 - 60- 40- 40- 20- 0+ 10 100 110 120 90 40 50 60 70 80 20 30 40 50 60 70 80 100 110 10 30 90 m/z m/z
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
In the first spectrum the base peak appears at M29 signifying ...View the full answer

Answered By

Jacob Festus
I am a professional Statistician and Project Research writer. I am looking forward to getting mostly statistical work including data management that is analysis, data entry using all the statistical software’s such as R Gui, R Studio, SPSS, STATA, and excel. I also have excellent knowledge of research and essay writing. I have previously worked in other Freelancing sites such as Uvocorp, Essay shark, Bluecorp and finally, decided to join the solution inn team to continue with my explicit work of helping dear clients and students achieve their Academic dreams. I deliver, quality and exceptional projects on time and capable of working under high pressure.
4.90+
1254+ Reviews
2845+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Below are mass spectra for four different compounds. Identify whether each of these compounds contains a bromine atom, a chlorine atom, or neither. a. b. c. d. 100- 80- 60- 60- 40 20- 0- 60 70 10 20...
-
The IR and mass spectra for three different compounds are shown in Figures 13.45-13.47. Identify each compound. a. b. c. 25 26 2.7 28 19 2000 100 43 58 100 71 85 0 20 40 60 80 100 120 m/z 13 14 15 6...
-
For each molecular formula, draw all the possible constitutional isomers of alcohols with that formula. Give the IUPAC name for each alcohol. (a) C3H8O (b) C4H10O (c) C3H6O (d) C3H4O
-
In a given week, 12 babies are born in hospital. Assume that this sample came from an underlying normal population. The length of each baby is routinely measured and is listed below (in cm): 49, 50,...
-
On July 1, 2013, Pizza Company decided to trade-in their used equipment (ovens, refrigerators, ect.) for new models at Sears. The old equipment was initially purchased for $120,000 in January 2010....
-
A long, thin, positively charged wire runs along the long central axis of a hollow conducting cylinder of length \(10 \mathrm{~m}\), inner radius \(50 \mathrm{~mm}\), and outer radius \(70...
-
Using a one-term Fourier cosine series, develop an empirical equation for solar declination as a function of day number counted from January 1 (see Table 5.1 and Figure 5.7). Figure 5.7 Table 5.1...
-
Day and Night formed an accounting partnership in 2008. Capital transactions for Day and Night during 2008 are as follows: Partnership net income for the year ended December 31, 2008; is $68,400...
-
Wa tch the above video and answer the following questions: https://www.youtube.com/watch?v=GoM4jIZbTtQ Should more video cameras be set up so that police can monitor and prevent criminal activity?...
-
1. Work up a buying plan to use when buying from Ghuman's. 2. How should Hughes distribute the allotted open-to-buy dollars among the available sizes, colors, and fabrics? Synopsis: A department...
-
Identify two peaks that are expected to appear in the mass spectrum of 3-pentanol. For each peak, identify the fragment associated with the peak, and show a mechanism for its formation.
-
How would you distinguish between each pair of compounds using high-resolution mass spectrometry? a. b. .
-
For any set A, finite or infinite, let B A be the set of all functions mapping A into the set B = {0, 1 }. Show that the cardinality of B A is the same as the cardinality of the set P(A). Then try to...
-
) Dividends Per Share Lightfoot Inc., a software development firm, has stock outstanding as follows: 25,000 shares of cumulative preferred 3% stock, $20 par, and 31,000 shares of $75 par common....
-
Discuss the impact of pharmacogenomics on personalized medicine and its implications for pharmacy practice.
-
Question 6 4 pts MEF Corporation had $600,000 in net income for the year ended December 31, 2018. What is Earnings per Share (in dollars and cents) if the company had 200,000 shares of common stock...
-
Blake Accounting pays Brooke Peet $93,600 per year. Assume that Blake's accountants are expected to work a total of 6,000 direct labor hours in 2024. Blake's estimated total indirect costs are...
-
A house owner is debating whether she should install drainpipes around her house to protect her house against heavy rains. The heavy rains tend to leak into the basement of the house. In the next...
-
Graph the solution set of the system of inequalities. - 2 3 ? 16 ?
-
If the joint cost function for two products is C(x, y) = xy2 + 1 dollars (a) Find the marginal cost (function) with respect to x. (b) Find the marginal cost with respect to y.
-
Predict the structure of the product of this reaction: The product has no infrared absorption in the 1620-1680-cm-1 region. NaOH in aqueous EtOH CI HS
-
Cis-4-Bromocyclohexanol racemic C6H10O (compound C) Compound C has infrared absorption in the 1620-1680-cm-1 and in the 3590-3650-cm-1 regions. Draw and label the (R) and (S) enantiomers of product C.
-
1-Bromo [2.2.1] bicycloheptane is unreactive toward both SN2 and SN1 reactions. Open the computer molecular model at the book's website titled "1-Bromo [2.2.1] bicycloheptane" and examine the...
-
The following data was extracted from the records of DT Ltd on 28 February 2021, the end of their financial year: R Share capital (900 000 shares at R2 par value) 1 800 000 Retained income 160 000...
-
THE FIRST ONE WITHOUT AN IDE AND THE SECOND ONE WITH AN IDE Task Description complete a Java application to draw different shapes with the specified color and size, subject to the following...
-
Suppose two factors are identified for the U.S. economy: the growth rate of industrial production, IP, and the inflation rate, IR. IP is expected to be 3% and IR 6%. A stock with a beta of 1 on IP...

Study smarter with the SolutionInn App


