The following compound has three nitrogen atoms: Each of the nitrogen atoms exhibits a lone pair that
Question:
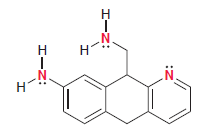
Each of the nitrogen atoms exhibits a lone pair that can function as a base (to abstract a proton from an acid). Rank these three nitrogen atoms in terms of increasing base strength using the following information:
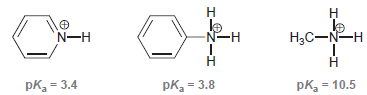
Transcribed Image Text:
:z エーZ: エーZ: Н N-H le Нас—N—н pk = 3.4 pk = 3.8 pk, = 10.5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
H IZ H ...View the full answer

Answered By

Joseph Ogoma
I have been working as a tutor for the last five years. I always help students to learn and understand concepts that appears challenging to them. I am always available 24/7 and I am a flexible person with the ability to handle a wide range of subjects.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following compound has two aromatic rings. Identify which ring is expected to be more reactive toward an electrophilic aromatic substitution reaction.
-
The following compound has only one asymmetric carbon. Why then does it have four stereoisomers? CH CH CHCH,CH CHCH Br
-
The following compound has been found to be an inhibitor of penicillinase. The enzyme can be reactivated by hydroxylamine (NH2OH). Propose a mechanism to account for the inhibition and for the...
-
The formula for the power output P of a battery is P = VI - RI where V is the electromotive force in volts, R is the resistance in ohms, and I is the current in amperes. Find the current that...
-
Northcutt Bikes purchases two types of wheels for their road racing bikes: a regular version and a deluxe version. The table below has demand information for the past year for each style of wheel, as...
-
The financial statements of Apple Inc. are presented in Appendix A. Instructions Refer to Apple's financial statements and answer the following questions. a. What were Apple's total assets at...
-
Following are observed frequencies. The null hypothesis is H0: p1 = 0.5, p2 = 0.3, p3 = 0.15, p4 = 0.05. a. Compute the expected frequencies. b. Compute the value of X2. c. How many degrees of...
-
Mike and Julie Bedard are a working couple. They will file a joint income tax return. This year, they have the following taxable income: 1. $125,000 from salary and wages (ordinary income) 2. $1,000...
-
A record company bought the rights to an artist's music catalogue and they expect to receive royalty payments of $30,000 per year forever (a perpetuity). What is this cash flow worth? Assume interest...
-
An analysis of the transactions made by Peat Deloitte & Co., a certified public accounting firm, for the month of August is shown below. The expenses were $560 for rent, $4,800 for salaries and...
-
For each pair of compounds below, identify the stronger base: (a) (b) (c) (d) (e) (f) -
-
Consider the following pKa values, and then answer the following questions: (a) For the following compound, will the lone pair on the nitrogen atom be more or less basic than the lone pair on the...
-
Can a leader influence others without power? How exactly would that influence take place?
-
Ruby Company manages the daily operations for the human resources department of Jade Company. In terms of the contract, Ruby receives a fee of \($100\) per year for each employee on Jades payroll,...
-
Rhodes Co. manages a school for the Old England School District. In year 1, Rhodes Co.s first year of operating the school, the school earned per pupil fees of \($100,000.\) Rhodes is responsible for...
-
Greasy Patties Corporation, a hamburger chain, has 100 stores. In its tenth year of operating, its total sales for year 10 amounted to \($100\) million. During year 11, Greasy Patties Corporation...
-
In the ____________case, the issue was that it did not accurately and completely describe the realities of its operations in its MD & A.
-
Explain the need for financial managers to balance liquidity and solvency.
-
How and why did the International Monetary Fund attempt to end beggar-thy-neighbor policies?
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
The radical reaction of propane with chlorine yields (in addition to more highly halogenated compounds)1-chloropropane and 2-chloropropane. Cl
-
In addition to more highly chlorinated products, chlorination of butane yields a mixture of compounds with the formula C4H9Cl. (a) Taking stereochemistry into account, how many different isomers with...
-
Chlorination of (R)-2-chlorobutane yields a mixture of dichloro isomers. (a) Taking into account stereochemistry, how many different isomers would you expect? Write their structures. (b) How many...
-
in accured interest, what is the formula and compute: a closing price of 101.375 on a particular day for IBM bond represents 101.3 percent of 1,000 or 1,0135 x $1,000
-
What is organizational structure? The arrangement or relationship of positions inside the organization The stable pattern of relationships in place to control and coordinate activities Organizational...
-
1.Describe the planning (planning) of the project and the components that must be be included in the planning process. 2. Describe the advantages and disadvantages of using a Gantt chart for project...

Study smarter with the SolutionInn App


