The following compound was isolated from nerve cells. (a) Describe how this compound differs in structure from
Question:
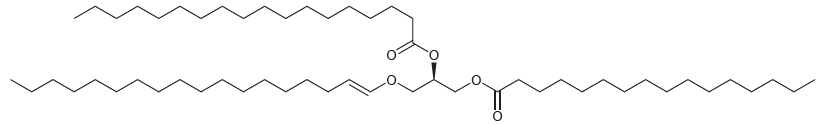
(a) Describe how this compound differs in structure from fats and oils.
(b) Three products are obtained when this compound is hydrolyzed with aqueous sodium hydroxide. Draw the structures of all three products.
(c) Four products are obtained when this compound is hydrolyzed with aqueous acid. Draw the structures of all four products.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a Fats and oils have a glycerol backbone connect...View the full answer

Answered By

Shayaan Moqueem
Currenntly , I am studying in an Engineering college named BIT Sindri . There is an NGO - " Prayaas India " which is run by the students of BIT Sindri and I have been a part of this since 2 years. The fundamental objective is to provide education to the children of the slum area near our college. They are basically belong to poor background. I have been teaching there since 2 years
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound was isolated as a minor constituent in an extract from garden cress. Its spectra are shown here. (1) Look at each spectrum individually, and list the structural characteristics you can...
-
Total RNA was isolated from nuclei of human cells growing in culture. This RNA was mixed with a purified, denatured DNA fragment that carried a large intron of a housekeeping gene (a gene expressed...
-
Compound W was isolated from a marine annelid commonly used in Japan as a fish bait, and it was shown to be the substance that gives this organism its observed toxicity to some insects that contact...
-
The author explores the dangerous world of undercover police work. Having read this chapter, do you believe this type of police work has the capability of corrupting police officers? If so why, and...
-
Superior Construction, Inc. is a home builder in Arizona. Superior uses a job order costing system in which each house is a job. Because it constructs houses, the company uses an account titled...
-
Use the equation (17.19) suggested by Pai for the turbulent stress and integrate for the velocity profile. How do the results compare with that of Prandtl? = 0.9835 H+ (1-#)['-(1-#)'] (17.19) H+
-
A manufacturer of smart phones has the following probability distribution for the number of defects per phone: (a) Determine the probability of 2 or more defects. (b) Is a randomly selected phone...
-
1. Compute he financial ratios discussed in the chapter for Wholesome Foods for 2012 and 2013. 2. Prepare a cash flow statement for the firm for 2012 and 2013.
-
After reading the Harvard Business Review article "Do You Know Your Cost of Capital?," why do you think financial managers make so many errors in estimating the components of the WACC, and what are...
-
Your client Kevins Train Spotting Limited holds a 25-year lease dated 01/01/2000 with 5 yearly reviews. The rent passing is currently 92,000 per annum. The Companys Director has approached you with a...
-
Starting with the following compound and using any other reagents of your choice, outline a synthesis for trimyristin. H.
-
Draw and name the polymer that results when each of the following monomers undergoes polymerization. (a) (b) (c) OAc c=C Vinyl acetate Br C=C Vinyl bromide
-
A Venturi meter is a device that is used for measuring the speed of a fluid within a pipe. The drawing shows a gas flowing at speed v2 through a horizontal section of pipe whose cross-sectional area...
-
Hannah manages a store for which she receives \(20 \%\) of any profit earned, so she does well when the store does well and earns nothing if the store incurs a loss. Hannah and the store's owner are...
-
Employees in the United Kingdom who are sick for at least four days in a row are entitled to statutory sick leave of \( 89.35\) per week for up to 28 weeks. Sick leave is paid by employers and is a...
-
Society uses various rules to decide which allocations are better than others. What rules does the generalized utilitarian welfare function use? If the generalized utilitarian welfare function were...
-
A man has a neighbor who owns a dog. The man has nothing against dogs as pets, but this one is yappy and barks incessantly. Let's say that the neighbor values the dog at its fair market value of \(...
-
In April 2011, the European Commission found Procter \& Gamble, Unilever, and Henkel, the leading producers of washing powder in Europe, guilty of operating a cartel from 2002 to 2005 in eight...
-
For the louvers shown in Fig. 16.21 and described in Problem 16.24, compute the torque required to rotate the louvers when the angle θ = 20°. Pivot Incoming air stream Louvers are...
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
NMR spectra for two compounds are given here, together with the molecular formulas. Each compound is a ketone or an aldehyde. In each case, show what characteristics of the spectrum imply the...
-
Show how you would accomplish the following syntheses. (a) Cyanohydrins acetorphenone cyanohydrin (b) Cyclopentanecarbaldehyde 2-cyclopentyl-2-hydroxyacetic acid (c) hexan-1-ol 2-hydroxyheptanoic...
-
Depending on the reaction conditions, two different imines of formula C8H9N might be formed by the reaction of benzaldehyde with methylamine. Explain, and give the structures of the two imines.
-
How do reviews and recommendations relate to situation-specific circumstances in the context of service evaluation?
-
You are performing an AFM measurement of CNTs on a surface. a) What is the "apparent" or "measured" radius (width) of a CNT of radius r if the radius of the AFM tip is R? Derive an algebraic...
-
During year-end planning session, they are considering another packaging supplier. 2023 annual sales projection would equate to 478,100 jars for their gourmet ketchup product. Purchase quantity Unit...

Study smarter with the SolutionInn App


