The following compounds cannot be made using only reactions that we learned in this chapter. For each
Question:
(a)
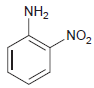
(b)
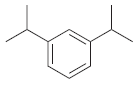
Transcribed Image Text:
NH2 „NO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a Nitration cannot be achieved effectively ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds cannot be prepared by an acetoacetic ester synthesis? Explain. (a) Phenyl acetone (b) Acetophenone (c) 3, 3-Dimethyl-2-butanone
-
Show how the following compounds could be prepared, using only the indicated starting material as the source of carbon: a. b. c. d. CH CH3CCH3 from CH3CHCH3 CH3 CH3CH-CCH3 O CH3 CH3C CHCH3 CH CH2CH
-
Show how the following compounds could be prepared, using only the indicated starting material as the source of carbon: a. b. c. d. e. f. OH HO
-
Simplify the expressions in Problems 3138. (3x - 1) (x + 3x - 2)
-
Which method of valuing a business is best? Why?
-
Briefly explain adjusting entries. List the four types of adjusting entries, and give an example of each type.
-
Data set: Daily calorie intakes (in kilojoules) of 28 people Construct a cumulative frequency distribution and an ogive for the data set using six classes. Then describe the location of the greatest...
-
Great Taste Food Stores operates 20 large supermarkets in the East. Each store is evaluated as a profit center, and store managers have complete control over their purchases and inventory policy....
-
2. Set up the following equations in the computer form: a. x+y=2+y-z 3z-1 b. 2xzy+x+ 4(3z+1)-y C. x = (y + z)
-
A particle with charge 7.80 C is moving with velocity u = - (3.80 X l03 m/s) j. The magnetic force on the particle is measured to be F = + (7.60 X l0-3N) i - (5.20 X l0-3N) k. (a) Calculate all the...
-
Using only reactions that we learned in this chapter, there are two different ways to prepare the following compound from benzene. Identify both ways, and then determine which way is likely to...
-
Starting with benzene and using any other necessary reagents of your choice, design a synthesis for each of the following compounds. In some cases, there may be more than one plausible answer. (a)...
-
An air-filled parallel-plate capacitor with plate separation d and plate area A is connected to a battery that applies a voltage V 0 between plates. With the battery left connected, the plates are...
-
Some companies agree that the move toward internationalization by U.S. universities primarily reflect an attempt to get more money because foreign buyers are willing to pay top dollar to obtain a...
-
Form teams of five members each. Each team represents a group of HR managers from a large petroleum company operating in a high-risk area. Some students are appointed as CEOs. Teams will role-play...
-
Research some recent articles dealing with the recent push by U.S. universities to start training foreign students abroad. Based on what you have read, what would you conclude in terms of the...
-
Do some research to learn why many multinationals prefer to send expatriates overseas rather than hire locals in dangerous areas. Based on your research, what are the advantages and disadvantages of...
-
A study was carried out on 60 electronic items to find the currents, x amperes, that could be safely passed through them at a fixed voltage before they overheat. The results are given in the two...
-
Give the equations of any vertical, horizontal, or oblique asymptotes for the graph of each rational function. x2 6 24 24 %3D f(x) = 5x2 26 + 5
-
Ann hires a nanny to watch her two children while she works at a local hospital. She pays the 19 year-old nanny $125 per week for 48 weeks during the current year. a. What is the employers portion of...
-
Identify the following two isomeric alkyl halides (C5H11Br) from their 300-MHz NMR spectra, which are as follows: Compound A: 0.91 (6/7, d, J = 6 Hz); 1.7-1.8 (3H, complex); 3.42 (2H, t, J = 6 Hz)...
-
Explain how the NMR spectra of 1, 2, 2-trimethyl-1-propanol would change following a D20 shake.
-
How would the NMR spectrum of ethyl fluoride differ from that of ethyl chloride?
-
The following partially complete T-accounts for the month of June along with additional information are from Renfrew & Company: Materials Inventory Work-in-Process Inventory Finished Goods Inventory...
-
The information that follows pertains to Julia Company: (a) Temporary differences for the year 2024 are summarized below. Expenses deducted in the tax return, but not included in the income...
-
Harwood Company uses a job-order costing system that applies overhead cost to jobs on the basis of machine-hours. The company's predetermined overhead rate of $2.60 per machine-hour was based on a...

Study smarter with the SolutionInn App


