The OH group on the side chain of serine is not deprotonated at a pH of 12.
Question:
Transcribed Image Text:
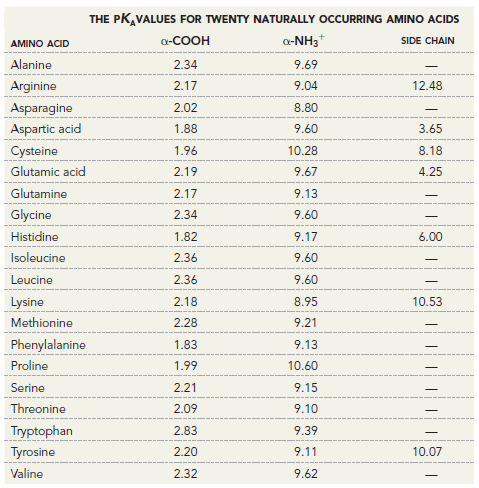
THE PK,VALUES FOR TWENTY NATURALLY OCCURRING AMINO ACIDS a-NH3* a-COOH SIDE CHAIN AMINO ACID Alanine 2.34 9.69 Arginine 2.17 9.04 12.48 Asparagine 2.02 8.80 Aspartic acid 1.88 9.60 3.65 Cysteine 1.96 10.28 8.18 Glutamic acid 9.67 2.19 4.25 Glutamine 9.13 2.17 Glycine 2.34 9.60 Histidine 9.17 1.82 6.00 Isoleucine 2.36 9.60 2.36 9.60 Leucine 8.95 Lysine 2.18 10.53 Methionine 9.21 2.28 Phenylalanine 1.83 9.13 Proline 10.60 1.99 Serine 2.21 9.15 Threonine 2.09 9.10 Tryptophan 2.83 9.39 Tyrosine 2.20 9.11 10.07 Valine 2.32 9.62 T|||||||
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
Tyrosine possesses a phenolic proton which is more readily depro...View the full answer

Answered By

Jane Thuita
I am a highly motivated and experienced tutor with a passion for helping students succeed. I have a strong background in a variety of academic subjects, including mathematics, science, and English. I have experience working with students of all ages and abilities, and I am able to customize my teaching methods to best fit each student's unique learning style. I am also able to work with students who have learning disabilities and those who are English language learners. I am patient, understanding, and dedicated to helping my students reach their full potential. In my free time, I enjoy reading, playing sports, and spending time with my family. I am excited to work with you and help you achieve your goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The citric acid cycle is a series of biological reactions that plays a central role in cell metabolism. The cycle includes dehydration reactions of both malic and citric acids, yielding fumaric and...
-
There are a number of conserved sequences found in an mRNA which dictate where splicing occurs. Where are these sequences found relative to the exon/intron junctions? What is the significance of...
-
Histidine possesses a basic side chain which is protonated at physiological pH. Identify which nitrogen atom in the side chain is protonated.
-
Once down to about 15, the worlds only wild flock of whooping cranes now numbers a record 237 birds in its Texas Coastal Bend wintering ground (www.SunHerald.com). The average whooping crane egg...
-
Marcel Company projects the following sales for the first three months of the year: $11,200 in January; $12,300 in February; and $11,100 in March. The company expects 60% of the sales to be cash and...
-
TQ 5G and Edge Computing 1- a retailer is able to track which products draw the most attention from its customers through the use of 5g-enabled motion sensors placed around the store. with this...
-
For Example 6.9, use a simulator to graph the effluent temperature of the methanol reactor as a function of the \(\mathrm{H}_{2} / \mathrm{CO}\) ratio. Data From Example 6.9:- Data From Example 6.8:-...
-
Kingston Company uses the dollar-value LIFO method of computing inventory. An external price index is used to convert ending inventory to base year. The company began operations on January 1, 2011,...
-
Write a MIPS assembly code with minimum possible assembly instructions for the following C++.. Assume that the variables num1, num2 and num3 are already saved in some MIPS registers. if (x > 0 && y...
-
Husband, who was suffering from schizophrenia and manic depression, had been in and out of a mental hospital, but at no time had he been declared insane by a court. In fact, he continued to work at...
-
Using the data in the following table, calculate the pI of the following amino acids. (a) Aspartic acid (b) Leucine (c) Lysine (d) Proline THE PK,VALUES FOR TWENTY NATURALLY OCCURRING AMINO ACIDS...
-
Explain why it is inappropriate to use a chiral catalyst in the preparation of glycine.
-
California Cement Co. produces its products two months in advance of anticipated sales and ships to warehouse centers the month before sale. The inventory safety stock is 20% of the anticipated...
-
Given that f(x)=x-2 and g(x)=5x+2, find (f+g)(2), if it exists. Select the correct choice below and, if necessary, fill in the answer box to complete your choice. OA. (f+g)(2)= (Simplify your...
-
Given the function g(y) = 3y + 11y, solve for g(y) = 4. Give an exact answer; do not round. (Use a comma to separate multiple solutions.) Provide your answer below: y =
-
how does 25000 of common stock issued at .98 instead of $1 par affect common stock outstanding of 100000
-
Rationalize the numerator and simplify. 11+2 7
-
Given a_(1)=6 and a common difference of -12, find the 18 th term in the sequence.
-
A fast ball thrown with a velocity of 40 m/s (approximately 90 MPH) is struck by a baseball bat, and a line drive comes back toward the pitcher with a velocity of 60 m/s. The ball is in contact with...
-
a. What is meant by the term tax haven? b. What are the desired characteristics for a country if it expects to be used as a tax haven? c. What are the advantages leading an MNE to use a tax haven...
-
In the addition of just 1 mole of bromine to 1 mole of hex-1-yne, should the hex-1-yne be added to a bromine solution or should the bromine be added to the hex-1-yne? Explain your answer.
-
Propose a mechanism for the entire reaction of pent-1-yne with 2 moles of HBr. Show why Markovnikov's rule should be observed in both the first and second additions of HBr.
-
Predict the major product(s) of the following reactions: (a) phenylacetylene + 2 HBr (b) hex-1-yne + 2 HBr (c) cyclooctyne + 2 HCl (d) hex-2-yne + 2 HCl + 2 HBr
-
Establishment Industries borrows $1,040 million at an interest rate of 7.2%. It expects to maintain this debt level into the far future. Establishment will pay tax at an effective rate of 38%. What...
-
The Presentation Layer ER Diagram shown in conjunction with part a below is focused around students who participate in sports and also enroll in classes. Please base your answer to this question on...
-
The current price of one share of XYZ stock is 56.50. A long straddle (one long call and one long put) on the stock with a strike price of K and one year to expiration has a total cost of 12.00. The...

Study smarter with the SolutionInn App


