Using acetophenone as your only source of carbon atoms, propose a synthesis for the following compound. Acetophenone
Question:
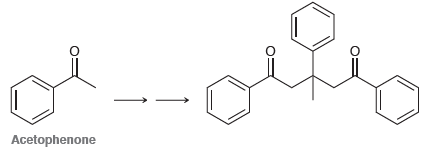
Transcribed Image Text:
Acetophenone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
RNH H HO ...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using benzene as your only source of carbon atoms and ammonia as your only source of nitrogen atoms, propose a synthesis for the following compound: HN NH
-
Using acetylene as your only source of carbon atoms, outline a synthesis for 3-hexyne.
-
Using acetylene as your only source of carbon atoms, design a synthesis of cis-3- decene:
-
During the year ended 30 June 20X5, Gem Stone Ltd acquired two areas of interest Site A and Site B. The exploration and evaluation activities at Site A amounted to $64454401. The costs relate to the...
-
An adjustable rate mortgage with a teaser start rate of 1.5%, an index of six month LIBOR, a margin of 2.0%, and periodic and lifetime caps of 2/6 with semi-annual adjustments, would have a maximum...
-
Show that the superposition of a radial velocity field and a torsional field results in a spiral flow. Sketch typical streamlines. Spiral flows are good prototypes for tornadoes. Explain why the...
-
True or False. A seismometer is also known as a vibrometer.
-
Flowmaster Forge Inc. is a designer and manufacturer of industrial air-handling equipment that is a wholly owned subsidiary of Howden Industrial Inc. Howden is interested in selling Flowmaster to an...
-
A Quincy man was arrested yesterday in connection with his alleged involvement in filing false tax returns on behalf of his company that hid over $10 million in corporate income. Su Nguyen, 60, was...
-
Richardson Ski Racing (RSR) sells equipment needed for downhill ski racing. One of RSRs products is fencing used on downhill courses. The fence product comes in 150-foot rolls and sells for $215 per...
-
Assign groups of students to go to an airport (or other location where they will find lots of foot traffic) to people watch. Have them classify as many people as possible according to social class...
-
Draw a complete mechanism for the following transformation. NaOH, heat
-
The graph of a function defined for > 0 is given. Complete the graph for , 0 to make (a) An even function and (b) An odd function. 4 0 x
-
The purpose of this assessment is to encourage the synthesis and integration of theoretical concepts with the context of answering a practical problem and offering commercial solutions to a client....
-
janine ( a single taxpayer ) has the following income tax related items in 2 0 2 3 : salary of $ 8 5 , 0 0 0 and for AGI deductions of $ 8 , 0 0 0 . Janine does not have any qualified business income...
-
What is A ^ ( 2 0 2 4 ) v , Where A = [ [ 1 , 3 ] , [ 2 , 2 ] ] and v = [ 0 , 5 ]
-
Q1 Saturn PLC Saturn PLC is considering a capital investment of 2,500,000 in new, state of the art equipment, with a resale value of 750,000 in 3 years' time. The equipment will be used to...
-
Common Pollution Policy grant of 20 per cent of gross capital expenditure is available, with payment in the year following expenditure. A disadvantage of the new equipment is that it will raise...
-
Find the volume of the following solids. The region bounded by y = 1/(x + 1), y = 0, x = 0, and x = 2 is revolved about the y-axis.
-
A non-charmed baryon has strangeness S = 2 and electric charge Q = 0. What are the possible values of its isospin I and of its third component I z ? What is it usually called if I = 1/2?
-
Give the principal organic product(s) expected when propionyl chloride reacts with each of the following reagents. (a) H2O (b) Ethanethiol, pyridine, 0 C (c) (CH3)3COH, pyridine (d) (CH3)2CuLi, -78...
-
Give the structure of a compound that satisfies each of the following criteria. (a) A compound C3H7ON that liberates ammonia on treatment with hot aqueous KOH (b) A compound that gives equal amounts...
-
When (R)-(-)-mandelic acid (-hydroxy--phenylacetic acid) is treated with CH3OH and H2SO4, and the resulting compound is treated with excess LiAlH4 in ether, then H2O, a levorotatory product is...
-
Doing a strategic analysis of GraceKennedy Limited, What is the current level of its economic performance, an indication of the factors responsible for the current performance and recommendations for...
-
How would you evaluate Cisco social media marketing success? Outline which, if any, of the mentioned media plans Cisco should pursue. Why? What are the challenges of using social media? Do you have...
-
Office Ltd manufactures furniture for computer work stations. The company uses a job costing system. Manufacturing overhead is applied to production at a predetermined overhead rate of $60 per direct...

Study smarter with the SolutionInn App


