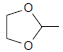
Using ethanol as your only source of carbon atoms, design a synthesis for the following compound:
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
HSO4 PCC ...View the full answer

Answered By

RISHABH YADAV
I have been tutoring since more than 2 years. I teach mathematics and physics. My students have qualified a number of examinations that have gained them jobs. Some of my friends regularly keep in touch with me so as to get their doubts cleared easily and with a friendly tone. My students have never complained of being late for classes or foe not considering their issues because I always keep my tutoring and my students at topmost priority.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
Using ethanol as your only source of carbon atoms, propose a synthesis for each of the following compounds. (a) (b) OEt .
-
Using toluene as your only source of carbon atoms, show how you would prepare the following compound.
-
Using acetylene as your only source of carbon atoms, design a synthesis of trans-5- decene:
-
Simplify each expression. Assume that all variables represent nonzero real numbers. (xy-3)-2
-
You are the Certified Managerial Accountant (CMA) for Northwood Company. You have worked for the company for several years and your responsibilities include working with managers to analyze business...
-
To begin to manufacture the VuYu, Bright Lights buys equipment from Crest Labs, Inc. Because Bright Lights does not have the funds to finance the purchase, Crest grants the buyer credit in exchange...
-
Lisa Sheraton, accountant for Chase Home Finance, was injured in a boating accident. Another employee prepared the accompanying income statement for the year ended December 31, 2008. The individual...
-
Hitachi, Ltd., reports total revenues of 9,041,071 million for its fiscal year ending March 31, 2013, and its March 31, 2013, unadjusted trial balance reports a debit balance for trade receivables...
-
Help please Kye Dewberry What are two questions you should be asking when assessing existing costs and resources? AABI Financial indicators: Non-financial indicators: GO PG
-
In the reverse-bias region the saturation current of a silicon diode is about 0.1 A (T = 20C). Determine its approximate value if the temperature is increased 40C.
-
Identify the starting materials needed to make each of the following acetals: (a) (b) (c) OEt
-
Propose an efficient synthesis for each of the following transformations: (a) (b)
-
Investors are vitally interested in a company's sales and profits, and its trends of sales and profits over time. Consider Foot Locker's sales and net income (net loss) during the period from 2005...
-
Consider a system described by the quasispin model of Problem 31.3 for $G>0$ with two identical fermions in a single $j$-shell. Show that the allowed seniorities are $v=0,2$, the allowed angular...
-
A point charge \(q=+5 \mathrm{nC}\) is at the center of a thick conducting spherical shell of internal radius \(10 \mathrm{~cm}\) and external radius \(12 \mathrm{~cm}\). (a) What is the magnitude...
-
Use the thermal-diffusivity value estimated from the above problem to find out whether any part of a fruit of diameter \(8 \mathrm{~cm}\) hanging in a tree will freeze if the ambient temperature...
-
At a point in a dryer benzene is evaporating from a solid. The air temperature is \(80^{\circ} \mathrm{C}\) and the pressure is \(1 \mathrm{~atm}\). The relative humidity of benzene in air is \(65...
-
An enterprise purchases an item of machinery on 1 April 2012 for ` 1,00,000. It depreciates this item at the rate of 10 per cent per annum on straight-line basis. On 1 April 2015, the enterprise...
-
Oakland Hills (OH) is a ski resort, and the revenue they report each year is greatly affected by the temperature and snowfall in the winter months. The variation in revenues from year to year is...
-
United Business Forms capital structure is as follows: Debt ............................................ 35% Preferred stock ........................... 15 Common equity .......................... 50...
-
Give the structure of each of the following compounds. (a) Chlorocyclopropane (b) Methylene iodide
-
Acetone (Table 8.2) has a significant dipole moment (2.7 D). Using structures, show the stabilizing interactions to be expected between acetone solvent molecules and (a) A dissolved potassium ion;...
-
Give the structure of A lecithin.
-
With a one day sale on Christmas Day and same day return policy in full effect, people filled the stores of Macy's to shop and return. The total revenue sales numbers were 2M with $150,000 in...
-
An ethical organizational culture creates an environment in which to structure behavior that is then evaluated by stakeholders. The key elements of an organizational culture include all of the...
-
Identify and discuss the seven (7) key dimensions of organizational culture Describe what ZenDesk's organizational culture looks like Discuss how ZenDesk uses its corporate culture to attain...

Study smarter with the SolutionInn App


