Valine has the structure Draw the zwitterion that would exist at neutral pH. H CH3CH-C-COOH - CH3
Question:
Valine has the structure
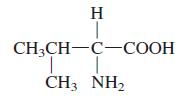
Draw the zwitterion that would exist at neutral pH.
Transcribed Image Text:
H CH3CH-C-COOH -СООН CH3 NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
Zwitterion ...View the full answer

Answered By

Himanshu Kumar
I have worked for various online sites as a doubt solver and subject experts .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Alanine has the structure Draw the zwitterion that would exist at neutral pH. CH3-C-COOH NH
-
Which one of the following five structures is a zwitterion? (a) (b) - O 2 CCH 2 CH 2 CO 2 - (c) H 3 N + CH 2 CO 2 - (d) CH 3 (CH 2 ) 16 CO 2 - K + (e) 0,CCH,CNH,
-
Consider the fully protonated amino acid valine: where the numbers denote the pKa values. (b) Calculate the predominant form of valine at pH 1.0, 7.0, and 12.0. (c) Calculate the isoelectric point of...
-
The top 5 stocks in the S&P 500 index, when ranked by market capitalization, make up 22% of the total market capitalization of the S&P 500 index. Numerical estimates of the mean (or expected) rates...
-
The number of automobile accidents reported per month helps to identify intersections that require improvement. The number of crashes per month reported at an intersection near a university campus in...
-
Refer to Exercise 78. a. Construct and interpret a 90% confidence interval for the true mean vertical jump (in inches) of the students at Haley, Jeff, and Nathans school. Assume that the conditions...
-
Consider the Michaelis-Menten model introduced in Eq. (12.23). Graph the expectation function for this model for \(\theta_{1}=200\) and \(\theta_{2}=0.04,0.06,0.08\), 0.10 . Overlay these curves on...
-
Kallus Corp. is an industry leader in the manufacture of toys. Each year, its design staff comes up with new ideas that are a great success. As a result, Kalluss sales and profits consistently exceed...
-
What are the mind boggling physiological components fundamental the body's pressure reaction, and how might a comprehension of these cycles illuminate progressed procedures for stress the executives?
-
The nuclear pore complex (NPC) creates a barrier to the free exchange of molecules between the nucleus and cytosol, but in a way that remains mysterious. In yeast, for example, the central pore of...
-
Consider the following polymer: From which two monomers is the polymer made? -C(CH2)10NH(CH2)6 NHC(CH2)10CNH(CH2)6NH-
-
If a sample of DNA isolated from a microorganism culture were analyzed and found to contain 1.5 mol of cytosine nucleotides and 0.5 mol of adenosine nucleotides, what would be the amounts of guanine...
-
How are metallic character and first ionization energy related?
-
List your experiences of sloppy digital marketing.
-
Using an approach similar to that in Worked Example 19.1, find all words with exactly one vowel (which might be repeated). What is the longest one? How many such words exist for each length? Data...
-
Why is the collection of the keys of a map a set and not a list?
-
What is the difference between a binary tree and a binary search tree? Give examples of each.
-
Consider an array list implementation with a removeLast method that shrinks the internal array to half of its size when it is at most half full. Give a sequence of addLast and removeLast calls that...
-
Malcolm owns 60% and Buddy owns 40% of Magpie Corporation. On July 1, 2015, each lends the corporation $30,000 at an annual interest rate of 10%. Malcolm and Buddy are not related. Both shareholders...
-
What is a make-or-buy decision?
-
A piston cylinder contains 3 kg of air at 20oC and 300 kPa. It is now heated up in a constant pressure process to 600 K.
-
An insulated cylinder is divided into two parts of 1 m3 each by an initially locked piston. Side A has air at 200 kPa, 300 K, and side B has air at 1.0 MPa, 1000 K. The piston is now unlocked so it...
-
A piston cylinder contains air at 600 kPa, 290 K and a volume of 0.01 m3. A constant pressure process gives 54 kJ of work out. Find the final temperature of the air and the heat transfer input.
-
Assume that a firm has weighted average cost of capital of 3.6% with a corporate tax rate of 21%. The pre-tax cost of debt is 3.1%. In case the firm would have no debt, the cost of equity for the...
-
Consider the stock of Aya Company, which will pay an annual dividend of $3 one year from today. The dividend will grow at a constant rate of 3%, forever.The market requires a 12% return on the...
-
Analyze the video presented on the link below. Also, please discuss or explain the video and relate it to personal finance. https://www.youtube.com/watch?v=nFY0HBkUm8o
Fault-Tolerance Techniques For High-Performance Computing 1st Edition - ISBN: 3319209434 - Free Book

Study smarter with the SolutionInn App


