We saw a general rule that the two protons of a CH 2 group will be chemically
Question:
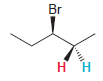
Transcribed Image Text:
Br нн Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The presence of the bromine atom does not render C 3 a chirality c...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
It is a general rule that any organic compound containing oxygen, nitrogen, or a multiple bond will dissolve in concentrated sulfuric acid. Explain the basis of this rule in terms of acid-base...
-
Two protons in an atomic nucleus repel each other, but they are also attracted to each other. Explain.
-
Two protons approach each other with different speeds. (a) Will the magnitude of the total momentum of the two-proton system be (1) greater than the magnitude of the momentum of either proton, (2)...
-
Consider Devine Fashion from S8-6. Assume that the fixed expenses assigned to each department include only direct fixed costs of the department (rather than unavoidable fixed costs as given in S8-6):...
-
What factors cause the differences in yields on U.S. government bonds?
-
A square clock of inertia \(m\) is hung on a nail driven into a wall (Figure P12.22). The length of each side of the square is \(\ell\), the thickness is \(w\), and the top back edge of the clock is...
-
Residual free energy is defined as (a) \(G^{R}=G-G^{\mathrm{ig}}\) (b) \(G^{R}=G^{\mathrm{ig}}-G\) (c) \(G^{R}=G+G^{\mathrm{ig}}\) (d) None of these.
-
Keairnes Supplies decided to change from LIFO to FIFO as of January 1, 2011. The change is being made for both book and tax purposes. 1. Using LIFO, the beginning retained earnings as of January 1,...
-
The IS-LM model is used to analyse simultaneous equilibrium in the goods market and the money market in the short run. You have the following information about an economy: C=50+0.8Yd (consumption...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
Identify the number of signals expected in the 1 H NMR spectrum of each of the following compounds. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) OMe MeO.
-
Identify the structure of a compound with molecular formula C 9 H 20 that exhibits four CH 2 groups, all of which are chemically equivalent. How many total signals would you expect in the 1 H NMR...
-
1. What changes should you suggest for the written exam? 2. What are the benefits of adding an essay to the written test? 3. How could you improve the interview portion of the hiring process? 4. Do...
-
You are evaluating an investment with a purchase price (outflow) of $100 today. However, you expect a $120 inflow at the end of each of the next 2 years. Assuming 5% nominal rate, compounded...
-
The cost of debt for firm XYZ is 6%.It's tax rate is 40%. The cost of retained earnings is 12% and the cost of external common equity is 14%. Retained earnings is $5000. The target capital structure...
-
The Marty's buy a house and borrow $350,000 from Rocket Mortgage Company. The mortgage carries an interest rate of 6% and is guaranteed for five years. After exactly two years of making monthly...
-
Solve the inequality:-n
-
Identify the y-intercept of f (x) = 23-7x23x+14 22+4x-7
-
Fill in the blank(s) to correctly complete each sentence. Factoring is the opposite of _________ .
-
Before the 1973 oil embargo and subsequent increases in the price of crude oil, gasoline usage in the United States had grown at a seasonally adjusted rate of 0.57 percent per month, with a standard...
-
Draw the structures of the following molecules: (a) Biacetyl, C4H6O2, a substance with the aroma of butter; it contains no rings or carboncarbon multiple bonds. (b) Ethylenimine, C2H5N, a substance...
-
Draw structures for the following: (a) 2-Methyiheptane (b) 4-Ethyl-2, 2-dimethylhexane (c) 4-Ethyl-3, 4-dimethyloctane (d) 2, 4, 4-Trimethylheptane (e) 3, 3-Diethyl-2, 5-dimethylnonane (f)...
-
Draw a compound that: (a) Has only primary and tertiary carbons (b) Has no secondary or tertiary carbons (c) Has four secondary carbons
-
Following are the issuances of stock transactions. 1. A corporation issued 8,000 shares of $20 par value common stock for $192,000 cash. 2. A corporation issued 4,000 shares of no-par common stock to...
-
A client calls in to sell their entire long position (300 shares) of AAPL and wanted you to walk them through the process. While going through BPROS you notice they are also short 3 calls contracts...
-
You expect an after-tax rate of return of 28% on your investments. A target company earns $263,000 (after taxes, per year). What is the maximum amount you would be willing to pay for this company?

Study smarter with the SolutionInn App


