What reagents would you use to perform each of the following transformations? a. b. H.
Question:
a.
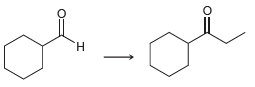
b.
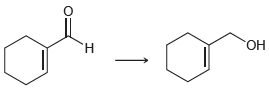
Transcribed Image Text:
H. н ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
a b H 1 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What reagents would you use to prepare the following compounds? a. b. CH CCH CH:C 0 CH3CCH-CH-CH (COCH-CH3 )2
-
What reagents would you use to prepare each of the following thiols: a. b. c. SH SH
-
What reagents would you use to bring about each step of the following syntheses? (a) (b) (c) (d) OH HiO4 (Section 22.6D) OH +NIN:
-
Assume a portfolio of two assets with the following total value (Al) covariance matrix 1 -0.5 -0.5 1 The sum of the 99% component VaRs of the two assets is:
-
Reids Company, which uses net present value to analyze investments, requires a 10% minimum rate of return. A staff assistant recently calculated a $500,000 machine's net present value to be $86,400,...
-
Describe which assets and capabilities were effectively leveraged by Nike for its digital growth.
-
To what degree is the current geographical distribution of wealth and power in the world today similar to what it was 1,000 years ago?
-
The partially completed inventory record for the table top subassembly in Figure shows gross requirements scheduled receipts, lead time, and current on-hand inventory. a. Complete the last three rows...
-
Silver Records has 4 employees who are paid on the 1 and 15 of each month for the work they performed in the preceding half-month. At November 30, each employee is owed gross pay of $2,250, but each...
-
Morn Ltd acquired 90% of the shares in Eve Ltd on 1 January 20X1 for £90,000 when Eve Ltds accumulated profits were £50,000. On 10 January 20X1 Morn Ltd received a dividend of...
-
A 1.25 mole sample of an ideal gas is expanded from 320. K and an initial pressure of 3.10 bar to a final pressure of 1.00 bar, and C P,m = 5/2R. Calculate w for the following two cases: a. The...
-
Propose a mechanism for the following reaction. NaH Br
-
Bromination of 3-methyl-2-butanone yielded two compounds, each having the molecular formula C5H9BrO, in a 95:5 ratio. The 1H NMR spectrum of the major isomer A was characterized by a doublet at 1.2...
-
You are a state-level health policy maker interested in health care access. You want to explore healthcare access barriers in underserved rural communities. You have contacts in these communities and...
-
Xavier is starting an online business called 'photobook' that is a social networking website where people post pictures of their books. Xavier has a lunch meeting with his sister Sally to discuss...
-
Describe some of the restrictions on the use of Termination for Convenience and the impact on the contractor.
-
The Holland Furnace Company's furnace repair scam involved several fraudulent tactics when dealing with consumers. What were these fraudulent tactics?
-
. Jason Jones is a flight attendant for Southeast Airlines. He went to Well-Mart (a large retail and pharmacy chain) for his COVID-19 vaccine. After he was fully vaccinated, he lost his paper vaccine...
-
Identify the type of conic section described. The conic section with eccentricity e = 0
-
Charles owns an office building and land that are used in his trade or business. The office building and land were acquired in 1978 for $800,000 and $100,000, respectively. During the current year,...
-
Predict the product of the reaction of valine with the following reagents: (a) CH3CH2OH, acid (b) Di-tert-butyl dicarbonate (c) KOH, H2O (d) CH3COCl, pyridine then H2O
-
Show how you could use the acetamidomalonate method to prepare the following amino acids: (a) Leucine (b) Tryptophan
-
Show how you could prepare the following amino acids using a reductive amination: (a) Methionine (b) Isoleucine
-
Excerpts from Andre Company's December 31, 2024 and 2023, financial statements are presented below: Accounts receivable Inventory Net sales Cost of goods sold Total assets Net income Total...
-
The York City Hospital has just acquired new equipment. The equipment cost $ 4 , 2 5 0 , 0 0 0 , and the organization spent $ 1 3 5 , 0 0 0 on upgrading the physical plant to the new equipment will...
-
Calculate the following showing all the necessary steps: Note: CPP rate to be used in the calculation is 5.95%. Note: Don't forget to deduct pay period exemption\ \ Tanya earns $25.00 per hour. This...

Study smarter with the SolutionInn App


