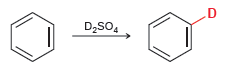
When benzene is treated with D 2 SO 4 , a deuterium atom replaces one of the
Question:
Transcribed Image Text:
D2SO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
H b8...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When benzene is treated with D2SO4, deuterium slowly replaces all six hydrogens in the aromatic ring. Explain.
-
When benzene is treated with propene and sulfuric acid (see structures of reactants below), two different monoalkylation products are possible. Draw their structures. Which one do you expect to be...
-
When benzene is treated with excess D2SO4 at room temperature, the hydrogens on the benzene ring are gradually replaced by deuterium. Write a mechanism that explains this observation.
-
Migration is a popular strategy among many species. Monarch butterflies migrate between the Sierra Madre mountains in Mexico and many locations across the USA and Canada. Answer the following...
-
Should a small business owner always purchase the products with the lowest prices? Why or why not?
-
Suppose that instead of the two moving blocks in Figure \(9.3 a\), just one block is used to compress the spring while the other end of the spring is held against a wall. (a) Is the system comprising...
-
Briefly discuss the use of cash registers, prelists, and daily cash summaries in controlling cash receipts.
-
Solar Designs is considering an investment in an expanded product line. Two possible types of expansion are being considered. After investigating the possible outcomes, the company made the estimates...
-
Issue 1 On 1 July 2021, we acquired an asset for $1,000,000, which it is depreciating using the straight-line method over 20 years (hence, $50,000 depreciation charged each year). We realised a...
-
A shaft with a circumferential groove is subjected to bending, as in Fig. A.12(c). The bending moment is M = 140 N m, and the dimensions are d 2 = 20, d 1 = 15, and p = 2.5 mm. It is made of a...
-
Calculate A o R and G o R for the reaction C 6 H 6 (l) + 15/2O 2 (g) 6CO 2 (g) + 3H 2 O(l) at 298 K from the combustion enthalpy of benzene and the entropies of the reactants and products.
-
A specialty fabrication shop has the following assembly processes for one of the products it makes: Process 1 feeds a completed component to Process 2; Process 2 feeds a completed assembly to Process...
-
The file MobileSpeed contains the overall download and upload speeds in mbps for nine carriers in the United States. Source: Data extracted from Best Mobile Network 2016, bit.ly/1KGPrMm, accessed...
-
If a PM has a -1% Fama-French alpha, would you expect this PM to underperform or outperform the S&P500 on average over time? Please explain carefully.
-
Portfolio (assume annual payments for each bond and mortgage) Maturity Type Annual Coupon/ Mortgage Rate Face Value of Holdings Par Value for each Bond 1 Treasury 0 100,000 100 5 Treasury 4% 100,000...
-
A car dealer offers you a car loan with payments of 6,000, semi-annually, for 3 years. If the posted APR on the loan is 3%, what is the cost of the car? Enter your response (rounded to 2 decimal...
-
Based on Intel Corporations annual report, how many offices in how many countries does in it operate in? How many employees do they have worldwide including its corporate office and company owned...
-
In the context of CAPM, if a company A has an expected rate of return of 2.5% and the consensus is that the market will grow by 3% and the risk free rate is at 0.2%, what can you say about the beta...
-
Use cylindrical or spherical coordinates, whichever seems more appropriate. A solid cylinder with constant density has base radius a and height h. (a) Find the moment of inertia of the cylinder about...
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
(a) Two isomeric S"2 products are possible when sodium thiosulfate is allowed to react with one equivalent of methyl iodide in methanol solution. Give the structures of the two oroducts. ...
-
When methyl iodide at 0.I M concentration is allowed to react with sodium ethoxide at 0.1 M concentration in ethanol solution, the product ethyl methyl ether is obtained in good yield. Explain why...
-
When methyl iodide at 0.I M concentration is allowed to react with sodium ethoxide at 0.1 M concentration in ethanol solution, the product ethyl methyl ether is obtained in good yield. Explain why...
-
American X wishes to borrow U.S. dollars at a fixed rate of interest. Corporation Y wishes to borrow Japanese Yen at a fixed rate of interest. The amounts required by the two companies are the same...
-
Bunga Raya Kuning Berhad is a Malaysian-based MNC that obtains 12 percent of its supplies from the U.K. manufacturers. Sixty-five percent of its revenues are from due to exports to U.K. where its...
-
Trevor's Tool Shop is considering investing in a new machine. The company currently has $500,000 per year in sales. The company has $265,000 per year in net income. If the company invests in the new...

Study smarter with the SolutionInn App


