When N,N-dimethylaniline is treated with bromine, ortho and para products are observed. Yet, when N,Ndimethylaniline is treated
Question:
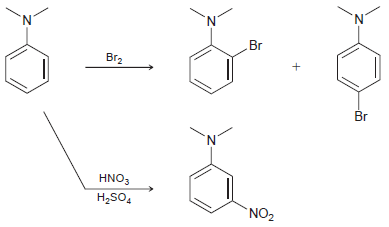
Transcribed Image Text:
Br Br2 Br N. HNO, H,SO, `NO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
The amino group in NNdimethylaniline is a strong activator and therefore an ort...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the product obtained when pyrrole is treated with a mixture of nitric acid and sulfuric acid at 0C.
-
The substitution reaction of toluene with Br2 can, in principle, lead to the formation of three isomeric bromotoluene products. In practice, however, only o-and p-bromotoluene are formed in...
-
Write resonance structures for the arenium ions formed when ethylbenzene reacts with a Br+ ion (as formed from Br2/FeBr3) to produce the following ortho and para products. Br FeBr Br
-
Mango Designs began selling its custom furniture on June 1, 2020. At the end of the month, the special journals showed the following results. Other information you will need is as follows: ? Interest...
-
Which of the following is not an argument against free trade? A) Trade keeps consumer prices too low. B) Small industries need protection to achieve comparative advantage in a global environment. C)...
-
Consider the following alternatives: i. \($150\) received in one year ii. \($220\) received in five years iii. \($330\) received in ten years a. Rank the alternatives from most valuable to least...
-
Suppose that \(v_{1}, v_{2}, \ldots, v_{n}\) are positive numbers. The arithmetic mean and the geometric mean of these numbers are, respectively, (a) It is always true that \(v_{A} \geq v_{G}\)....
-
A country currently imports automobiles at $8,000 each. Its government believes that, given time, domestic producers could manufacture autos for only $6,000 but that there would be an initial...
-
Cost of common stock equity Ross Textiles wishes to measure its cost of common stock equity. The firm's stock is curently seling for $4053. The firm just recently paid a dividend of 54.01. The fient...
-
Show that X62 can be computed with only eight multiplications.
-
Propose a plausible mechanism for the following transformation: . . H,SO.
-
Assign a systematic (IUPAC) name to each of the following compounds: (a) (b) (c) (d) (e) Br Br
-
a. What is a captive insurer? b. Explain the advantages of a captive insurer in a risk management program.
-
On December 31, Year 4, Rent Company exchanged 25,000 shares of its $10 par value stock for all of the outstanding shares of Subic Company. The fair value of Rent's stock was $40 at the date of...
-
Wales's age 644 has been part if his employer dbl for the last 26 years. Pension contributions are 4% of his salary and this is matched by his employer benefits under the plan are based on 1.5% of...
-
To continue the balance sheet workings, total liabilities 161,500; under the stockholders' equity: common stock is 5,000; additional paid-in capital is 15,000; retained earnings is missing; total...
-
An electron initially moves left to right long the x axis with a KE of 3.33 x 10-17 J. It moves into an electric field, which points in the positive x direction, and travels a distance of 0.600 m...
-
A measurement of an electron's speed is 2.0 x 106 m/s and has an uncertainty of 10%. What is the minimum uncertainty in its position? (h = 6.626 x 10 34 J s, mel 9.11 x 1031 kg)
-
The results of a medical test show that of 85 people selected at random who were given the test, 80 tested negative and 5 tested positive. Determine the odds against a person selected at random from...
-
Give the products of the following reaction, where T is tritium: dioldehydrase Ad- CH CH3C-COH CoIII) coenzyme B12
-
Write three-dimensional (wedge-dashed wedge-line) representations for each of the following: (a) CH3Cl (b) CH2Cl2 (c) CH2BrCl (d) CH3CH2Cl
-
(a) Write two resonance structures for the formate ion HCO2-.(The hydrogen and oxygen atoms are bonded to the carbon.) (b) Explain what these structures predict for the carbon-oxygen bond lengths of...
-
Write the resonance structure that would result from moving the electrons as the curved arrows indicate. Be sure to include formal charges if needed. (a) (b) (c) (d) 079 HIN:
-
While taking the measurement of a different sphere with a radius of r = 2 . 2 2 cm , your instrument has a measurement uncertainty of \ pm 0 . 0 4 cm . What is the propagated uncertainty of the...
-
A sphere slides along a friction-free inclined surface n. The sphere slides from a height of n for the speed of the sphere once it reaches the point2. (b) 4.0m. (a) Develop an expression What is...
-
10.21 Estimate the hydrogen-burning lifetimes of stars near the lower and upper ends of the main sequence. The lower end of the main sequence 2 occurs near 0.072 Mo, with log10 Te = 3.23 and log 10...

Study smarter with the SolutionInn App


