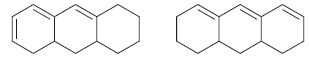
Which of the following compounds below do you expect to have a longer λ max ?
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Two of the bonds are ...View the full answer

Answered By

Sheikh Muhammad Ibrahim
During the course of my study, I have worked as a private tutor. I have taught Maths and Physics to O'Level and A'Level students, as well as I have also taught basic engineering courses to my juniors in the university. Engineering intrigues me alot because it a world full of ideas. I have passionately taught students and this made me learn alot. Teaching algebra and basic calculus, from the very basics of it made me very patient. Therefore, I know many tricks to make your work easier for you. I believe that every student has a potential to work himself. I am just here to polish your skills. I am a bright student in my university. My juniors are always happy from me because I help in their assignments and they are never late.
4.90+
14+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds would you expect to exhibit only London forces? a. Potassium chloride, KCl b. c. Silicon tetrafluoride, SiF4 d. Phosphorus pentachloride, PCl5 (
-
Which of the following compounds would you expect to be the most generally reactive, and why? DO
-
Which of the following compounds could be resolved into enantiomers at room temperature? Explain. (a) (b) CH2CH CH3)2 CH CH CH3 CH3
-
You mentioned that Amazon has sustainability topics within us supply chain. How we deal with the criticism that Amazon itself tribute to overconsumption that's antithetical to sustainability
-
The figure shows a proposed site where an excavation will be made. The 10 ft layer of sand will be removed, so that the top of the 24 ft. normally consolidated clay layer will be exposed. Assume full...
-
Sam Jones owns a granite stone quarry. When he acquired the land, Sam allocated $800,000 of the purchase price to the quarry's recoverable mineral reserves, which were estimated at 10 million tons of...
-
A diesel engine has a compression ratio of 20 and cut-off takes place at 5\% of the stroke. Find the air-standard efficiency. Assume \(\gamma=1.4\).
-
Suppose a credit market with a good borrowers and 1-a bad borrowers. The good borrowers are all identical, and always repay their loans. Bad borrowers never repay their loans. Banks issue deposits...
-
Based on an aging of accounts receivables, management assigned 1% to the $100,000 of receivables 0-30 days outstanding, 5% to the $10,000 receivables 31-60 days and 20% to the $1,000 of receivables...
-
Target adopted the new leasing standard for the year ended February 2, 2019, using the modified retrospective approach outlined in ASC Topic 842. All questions relate to the year ended February 2,...
-
Suppose that there are 1 million federal workers at the lowest level of the federal bureaucracy and that above them there are multiple layers of supervisors and supervisors-of-supervisors. Assume...
-
Give arguments to support both the relevance and the irrelevance of paying dividends.
-
Suppose real GDP is $10,000 billion and the basic expenditure multiplier is 2. If two tax changes are made at the same time: a. fixed taxes are raised by $100 billion, b. the income-tax rate is...
-
John Doe had earned commissions on life insurance sold prior to his death. The commissions of $20,000 were to be paid in five annual installments. The executor of his estate collected the first...
-
An estate with depreciable property may not do one of the following: a. Pay income to first-tier beneficiaries b. Pay income to second-tier beneficiaries c. Distribute capital losses on termination...
-
Zap Industries had a tentative general business credit in 2019 of $60,000. Its tax liability in 2018 was $20,000. Its tax liability before the general business credit in 2019 is $50,000. a. What is...
-
The following court cases have helped to clarify the rightful recipient of depreciation of property held by an estate or trust. Read the following judicial decisions and prepare a brief written...
-
Yoyo Corporation bought $42,000 worth of energy equipment in November 2014, taking an investment credit of $4,200. In March 2019, the firm sells the equipment for $30,000. What is the investment...
-
Open the Beachwood Solution.sln file contained in the VB2017\AppC\Beachwood Solution folder. Use what you learned in the appendix to debug the application.
-
You've been asked to take over leadership of a group of paralegals that once had a reputation for being a tight-knit, supportive team, but you quickly figure out that this team is in danger of...
-
Which, if any, of the following compounds can be prepared by an acetoacetic ester synthesis?Explain. (a) Br. (c) (b) CH CH - CH
-
How would you prepare the following ketones using an acetoacetic estersynthesis? (a) (b) CH-H CH-CH CH2CH2CHCH3 CH3 CH
-
How would you prepare the following compounds using either an acetoacetic ester synthesis or a malonic estersynthesis? (d) (c) ( (a) H CHO2Et H H2H,H3 "CH Et
-
1. How does marketing create value, and how do firms become more value driven? 2. Describe a firm's stakeholders and how they are impacted by conscious marketing.
-
Beginning inventory 100 units @ $8.00 = $ 800 Purchase # 1 200 units @ $6.00 = 1,200 Purchase # 2 100 units @ $12.00 = 1,200 Total 400 units $3,200 Ending inventory is 150 units. 1. What is cost of...
-
Sam's Ice Creams, a cottage country favourite in Northern Ontario, has launched a new range of ice creams branded as "Summer Flavours." To capitalize on the cottage and tourism season from May to...

Study smarter with the SolutionInn App


