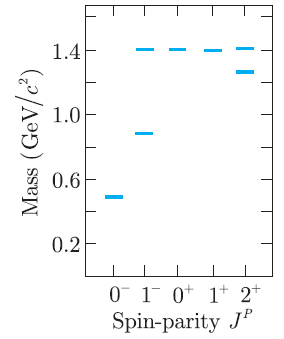
The K + and its excited states with masses below 1.5 GeV/c 2 are shown in Figure
Question:
Figure 3.12
Transcribed Image Text:
1.4 1.0- 0.6 0.2- 0 1 0* 1+ 2* Spin-parity J" Mass (GeV/c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
The possible spectroscopic states and their J P values are the same as those ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
The first excited state of Ca is reached by absorption of 422.7-nm light. (a) Find the energy difference (kJ/mol) between ground and excited states. (b) The degeneracies are g*/g0 = 3 for Ca. Find...
-
Gold, 19879Au, undergoes decay to an excited state of 19880g. H the excited state decays by emission of a photon with energy 0.412MeV, what is the maximum kinetic energy of the electron emitted in...
-
Triplet excited states some organic molecules have a triplet (S = 1) excited state at an energy k B above a singlet (S = 0) ground state. (a) Find an expression for the magnetic moment () in a field...
-
Researchers at the Hospital for Sick Children in Toronto compared babies born to mothers with diabetes to babies born to mothers without diabetes (Conditioning and Hyperanalgesia in Newborns Exposed...
-
On January 1, 2017, the ledger of Russell Software contains the following liability accounts. Accounts Payable..........................42,500 Sales Taxes Payable..........................5,800...
-
Complete the following table describing the short-run daily costs of the Attractive Magnet Co. for2009. Total Total Costs Total Costs Fixed Costs Costs Total Costs Costs S 30 Average Product Total...
-
When is it appropriate to seek a writ of possession as a provisional remedy?
-
Contrast the arguments concerning union membership that are likely to be presented by a union with those likely to be presented by an employer.
-
When most people think about how difficult college is, they naturally think about coursework. Few people, however, are aware of the challenges that can accompany academic life. What are the...
-
You have been employed as a consultant to Golf R Us, Inc. The company has been having problems with account receivables and they want to use various dunning methods to better their receivables. But...
-
If pp annihilation at rest proceeds via S-states, explain why the reaction pp 0 0 is forbidden as a strong or electromagnetic interaction.
-
Which of the following six decays are allowed in lowest-order weak interactions? (a) K + + + + + e + e , (b) K + + + e + e , (c) 0 + e + + e , (d) 0 + e + v e , (e) 0 p + + ...
-
Is the anti-takings provision of the ESA constitutional?
-
What four types of financial statements does the annual report typically include?
-
Indicate whether each of the following statements is true or false by writing T or F i n t he a nswer c olumn. Slander is a spoken, and libel is a written, form of defamation.
-
To what types of risk are investors of foreign bonds exposed?
-
Indicate whether each of the following statements is true or false by writing T or F i n t he a nswer c olumn. Offshore gambling refers to foreign citizens placing bets in casinos located within the...
-
What is the statement of stockholders equity, and what information does it provide?
-
For the cash flows shown, determine the future worth in year 5. Cash Flow, $/Year Estimated / Per Year 12% 12% 20% Year 5000 6000 1-4 9000
-
The unadjusted trial balance of Secretarial Services is as follows: SECRETARIAL SERVICES Unadjusted Trial Balance as at 31 December 2017 Account Debit Credit Cash at bank Office supplies Prepaid...
-
Solve Problems A.1 through A.6 using matrices [A], [B], [C], [D], and [E] given by (a) |A| + |B| (b) |A| + |B| (c) |A| |C| T (d) |D| {B} (e) |D| |C| (f) |C| |D| 3 1 01 -1 0 3 2 0 [4] [B] [C] -2 8 3 1...
-
Breathing Oxygen. The density of air under standard laboratory conditions 1.29 kg/m 3 is and about 20% of that air consists of oxygen. Typically, people breathe about L of air per breath. (a) How...
-
(a) The recommended daily allowance (RDA) of the trace metal magnesium is 410 mg/day for males. Express this quantity in g/day. (b) For adults, the RDA of the amino acid lysine is 12 mg per kg of...
-
A photoelectron is produced from a bismuth surface (which has a work function of 4.31 eV) at a speed of 0.910c. Relativistic effects should be included . What wavelength of light was used to produce...
-
6. Decarburization is the decrease of the carbon content of (the surface of) a steel due to interactions with the environment at elevated temperatures. This will result in a layer on the surface with...
-
Discussion Topic Activity Time: 3 Hours Directions: Think of the company and the industry you currently work for. (If you aren't currently working, consider your previous employer.) Discuss how your...

Study smarter with the SolutionInn App


