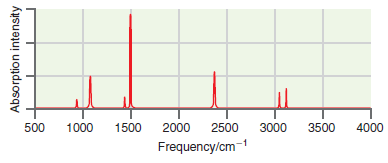
A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use
Question:
Transcribed Image Text:
500 1500 1000 2000 2500 3000 3500 4000 Frequency/cm-1 Absorption intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
The peaks near 3100 cm 1 are indicative of CH stretching modes and the pea...View the full answer

Answered By

Emel Khan
I have the ability to effectively communicate and demonstrate concepts to students. Through my practical application of the subject required, I am able to provide real-world examples and clarify complex ideas. This helps students to better understand and retain the information, leading to improved performance and confidence in their abilities. Additionally, my hands-on approach allows for interactive lessons and personalized instruction, catering to the individual needs and learning styles of each student.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A simulated infrared absorption spectrum of a gas phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
The investment shown in the following figure has an annual equivalent worth of $200 at i = 8%. Determine the cash flows in periods 2, 3, 5 and 6. $400 $400 3 4 5 6. Years $2,145
-
A tensile testing machine is shown in the following figure to determine the behavior of materials as they are deformed. (a) Use the provided data to calculate the stress and the corresponding strain...
-
A physical pendulum of mass m = 3 . 3 6 kg is comprised of an odd shape that has a centre - of - mass a distance of d = 0 . 5 5 5 m from the pivot point. The pendulum is displaced from equilibrium to...
-
In Exercises 1-4, use a graphing utility to approximate the solutions of the equation in the interval [0, 2). 1. cos(x + / 4) + cos(x - / 4) = 1 2. tan(x + ) - cos(x + / 2) = 0 3. sin(x + / 2) +...
-
A manufacturing process produces 500 parts per hour. A sample part is selected about every half hour, and after five parts are obtained, the average of these five measurements is plotted on an x...
-
A panel consists of three female and seven male experts. Three experts are chosen at random from this panel to serve in a selection committee. What is the probability of choosing (a) three men? (b)...
-
Refer to the Creative Technology example on delaying write-downs of current assets. How much excess inventory do you estimate Creative Technology is holding in March 2005 if the firms optimal days...
-
Your supervising attorney, Lillian Liberty, is responsible for conducting a professional training seminar for all the firm's paralegals. Liberty has asked you to prepare "answers" to several...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
Isotopic substitution is used to identify characteristic groups in an unknown compound using vibrational spectroscopy. Consider the C~C bond in ethane ( 12 C 2 1 H 6 ). By what factor would the...
-
The fundamental vibrational frequencies for 1 H 2 and 2 D 2 are 4401 and 3115 cm 1 , respectively, and De for both molecules is 7.667 10 19 J. Using this information, calculate the bond energy of...
-
What special measures might be necessary for a successful diversity-oriented recruiting effort?
-
There are computers in Internet cafes, at conferences, and in airport lounges that should be considered unsafe for any personal use. Revise the following sentences to avoid unnecessary there is/are...
-
Electrostatic work \(W\) is done on a charged particle as the particle travels from point \(A\) to point \(B\) in an electric field. You then apply a force to move the particle back to A, increasing...
-
A positively charged particle initially at rest on the ground accelerates upward to \(100 \mathrm{~m} / \mathrm{s}\) in \(2.00 \mathrm{~s}\). If the particle has a charge-to-mass ratio of \(0.100...
-
The Plant Assets account and Accumulated DepreciationPlant Assets account of Star Media show the following: Star Media sold plant assets at an $11,000 loss. Where on the statement of cash flows...
-
A small, charged, spherical object at the origin of a Cartesian coordinate system contains \(3.30 \times 10^{4}\) more electrons than protons. What are the magnitude and direction of the electric...
-
Find the derivatives of the following functions. y = 4 -x sin x
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
The half-life (t 1/2 ) of a radioactive species is the time it takes for half of the species to emit radiation and decay (turn into a different species). If a quantity N 0 of the species is present...
-
Perform the following pressure conversions, assuming when necessary that atmospheric pressure is 1 atm. Unless otherwise stated, the given pressures are absolute. (a) 2600 mm Hg to psi (b) 275 ft H 2...
-
The viewing window in a diving suit has an area of roughly 65 cm 2 . (a) If an attempt were made to maintain the pressure on the inside of the suit at 1 atm, what force (N and lb f ) would the window...
-
November 2021 (actual) December 2021 (budgeted) January 2022 (budgeted) Cash sales $ 80.000 Credit sales Total sales 240,000 $320,000 $100,000 360,000 $460,000 $ 60,000 180,000 $240,000 Management...
-
A firm has 200 shareholders, you among them. Each shareholder owns $20 worth of sto addition, Mr. Hostile owns 50 shares (for a firm total of 250 shares) and is trying to fi management. In an attempt...
-
You own a Triplex and are renting each unit for $1029/month Expenses will be 45% of gross rent Vacancy rate is 5% You will also have additional income by charging $25/month/unit for each unit's use...

Study smarter with the SolutionInn App


