(a) Two (unnormalized) excited state wavefunctions of the H atom are Normalize both functions to 1. (b)...
Question:
(a) Two (unnormalized) excited state wavefunctions of the H atom are
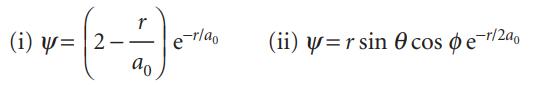
Normalize both functions to 1.
(b) Confirm that these two functions are mutually orthogonal.
Transcribed Image Text:
(i) u = 2 ਹੈ % -r/ao (ii) y=r sin cos e-r/2a0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What are the units of the H atom total energy eigenfunctions? Why is a 0 3/2 R(r) graphed in Figure 20.6 rather than R(r)?
-
A hydrogen atom is excited from its ground state to the state with n = 4. (a) How much energy must be absorbed by the atom? Consider the photon energies that can be emitted by the atom as it...
-
Two unnormalized functions of the hydrogen atoms are (2)2 1 1 2 2 sincos 2 Where N1 and N2 are the normalization constants The volume element for spherical polar coordinate is 2 sin and the limits...
-
Neutrons can be used in diffraction experiments to probe the lattice structure of crystalline solids. Since the neutron's wavelength needs to be on the order of the spacing between atoms in the...
-
You are given two paired samples with the following information: Based on these paired samples, test at the = 0.05 level whether the true median paired difference is 0. 2 le 3 4 O 2 1 5 9 438...
-
Two stationary point charges +3.00 nC and +2.00 nC are separated by a distance of 50.0 cm. An electron is released from rest at a point midway between the two charges and moves along the line...
-
When is it appropriate to seek a writ of possession as a provisional remedy?
-
The American Express Company is a major financial services company, noted for its American Express card. Below are some of the performance measures used by the company in its balanced scorecard....
-
Indian Limited has an established accounting practice of allocating overhead costs using an allocation base of direct labor dollars. Indian limited also has an established accounting practice of...
-
Darcelle teaches skiing at the Snow School. Jack runs a ski shop called Up Hill, Down Hill. Their combined bank interest is $1,000. If they get a refund, they would like the money deposited into...
-
Determine which of the following functions are eigenfunctions of the inversion operator (which has the effect of making the replacement x x): (a) X 3 kx, (b) Cos kx, (c) X 2 + 3x 1. State the...
-
Calculate the average linear momentum of a particle described by the following wavefunctions: (a) E ikx , (b) Cos kx, (c) E x2 , where in each one x ranges from to +.
-
Russell (birthdate February 2, 1966) and Linda (birthdate August 30, 1971) Long have brought you the following information regarding their income and expenses for the current year. Russell owns and...
-
Massi unlimited is interested in hiring you as a Hidden. You started a management accountant in your final job. Interview question came from the chief financial officer who asked you how activity...
-
Carry out an essay focused on answering and substantiating the following questions: What is an account and how does it help the registration process? Explain the accounting recording process. What...
-
(a) The grand partition function for a single particle state in a system of bosons is given by: Z = eBlu-e, jn, Discuss the constraint on the chemical potential u, and show that, provided that this...
-
Dr. Helen Fisher is a Biological Anthropologist and the Scientific Advisor to Match.com. She has conducted extensive research and written 6 books on the evolution and future of human sex, love,...
-
Total sales were booked to the sales account without accounting for sales tax collected. What is the correct journal entry to record sales tax collected? When analyzing financial reports, such as the...
-
Do you think the cows mind?
-
The words without recourse on an indorsement means the indorser is: a. not liable for any problems associated with the instrument. b. not liable if the instrument is dishonored. c. liable personally...
-
Verify that the radius ratio for (a) sixfold coordination is 0.414, and (b) for eightfold coordination is 0.732.
-
In an X-ray investigation, the following structure factors were determined (with F h00 =F h00 ): Construct the electron density along the corresponding direction. h F1,00 0 10 1 -10 2 8 3 -8 4 6 5 -6...
-
What are the values of the angle of the first three diffraction lines of bcc iron (atomic radius 126pm) when the X-ray wavelength is 72pm?
-
A regional electrical distributor currently has 1,000 customers who buy an average of $5,000 per year, generating a 50% margin. From experience, the company knows that 20% of its customers will not...
-
Suppose you observed that one-year T-bills are trading with a yield to maturity (YTM) of 4.75%. The yield spread between AAA and BB rated corporate bonds is 130 basis points. The maturity yield...
-
You have $36,000 to Invest in Sophie Shoes, a stock selling for $60 a share. The initial margin requirement is 65 percent. calculate your rates of return if the stock rises to $90 a share and if it...

Study smarter with the SolutionInn App


