At 300 K, the partial vapour pressures of HCl (that is, the partial pressure of the HCl
Question:
At 300 K, the partial vapour pressures of HCl (that is, the partial pressure of the HCl vapour) in liquid GeCl4 are as follows:
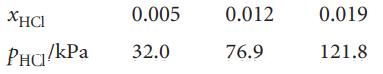
Show that the solution obeys Henry’s law in this range of mole fractions, and calculate Henry’s law constant at 300 K.
Transcribed Image Text:
ХНСI PHO/kPa 0.005 32.0 0.012 76.9 0.019 121.8
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Henrys law states that the partial pressure of a gas in a liquid is directly propor...View the full answer

Answered By

Dansteve Matoke
As a consequence of more than three years of experience in ACADEMIC WRITING, I have vast, diverse knowledge and impeccable grammar. I guarantee the quality of my work across multiple fields. I intend to precisely meet the clients' expectations. Let's work together to achieve exceptional grades.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At 310 K, the partial vapour pressures of a substance B dissolved in a liquid A are as follows: xB 0.010 0.015 0.020 Pg/kPa 82.0 122.0 166.1 Show that the solution obeys Henry's law in this range of...
-
At 300 K the reaction below obeys the rate law rate=k[NOCl] 2 where k= 2.8x10 -5 M -1 * S -1 2NOCl ------> 2NO + Cl 2 Suppose 1.0 mole of NOCl is introduced into a 2.0- liter container at 300 k....
-
The vapour pressure of pure liquid A at 293 K is 68.8 kPa and that of pure liquid B is 82.1 kPa. These two compounds form ideal liquid and gaseous mixtures. Consider the equilibrium composition of a...
-
Data Set 32 "Airport Data Speeds" in Appendix B includes Sprint data speeds (mbps). The accompanying TI-83 / 84 Plus display results from using those data to test the claim that they are from a...
-
You bought a stock for $40, received a dividend of $1, and sold it for $41 after five months. What is your annualized arithmetic rate of return?
-
A TS-Diagram (a) Graph a Carnot cycle, plotting Kelvin temperature vertically and entropy horizontally. This is called a temperature-entropy diagram, or TS-diagram. (b) Show that the area under any...
-
A simple random sample of 75 people are given a new drug that is designed to relieve pain. After taking this drug for a month, they switch to a standard drug. The question of interest is whether the...
-
Prepare budgetary entries, using general ledger accounts only, for each of the following unrelated situations: a. Anticipated revenues are $10 million; anticipated expenditures and encumbrances are...
-
10 Suppose a mutual fund manager has been investing in value stocks. Given the historical patterns in stock returns in the U.S., would the fund manager prefer to be evaluated by having alpha...
-
The base of the square foundation, 3 mx 3 m, is located at 1 m depth. The soil supporting foundation has a friction angle of '= 20 and c'= 20 kN/m. The unit weight of soil, y, is 21 kN/m. What is the...
-
How is Raoults law modified so as to describe the vapour pressure of real solutions?
-
The partial molar volumes of acetone (propanone) and chloroform (trichloromethane) in a mixture in which the mole fraction of CHCl 3 is 0.4693 are 74.166 cm 3 mol 1 and 80.235 cm 3 mol 1 ,...
-
The cantilevered beam is subjected to a couple moment \(\mathbf{M}_{0}\) applied at its end. Determine the slope of the beam at \(B\). \(E I\) is constant. Use the method of virtual work. A -L B Mo
-
Michelle wants to compare vulnerabilities she has discovered in her data center based on how exploitable they are, if exploit code exists, and how hard they are to remediate. What scoring system...
-
A marketing analyst is asked to build a simulation for a new product launch. The new product's net profit= sales volume * (sales price - unit cost) - fixed costs The analyst's market research has...
-
Suppose one of GP Manufacturings executives typically uses the payback as a primary capital budgeting decision tool and wants some payback information. a. What is the projects payback period? b. What...
-
JR has recently witnessed his friend Beth's success with her real estate investments, and he would like to pursue an investment of his own. JR has discovered an investment property that will provide...
-
A car can travel faster when the of a road is smaller. Let's assume that the speed around a curve is to the square root of the curvature. A car is moving along the path given by f ( x ) = x ( these...
-
What aspects of the bank's service quality specification have been revealed to the customer? Are these reasonable for such an account?
-
Using Apple, demonstrate how the differentiation strategy can be well implemented.
-
Given that F=dV/dr, calculate the distance dependence of the force acting between two non-bonded groups of atoms in a polymer chain that have a London dispersion interaction with each other.
-
D.D. Nelson et al. (Science 238, 1670 (1987)) examined several weakly bound gas-phase complexes of ammonia in search of examples in which the H atoms in NH 3 formed hydrogen bonds, but found none....
-
Estimate the energy of the dispersion interaction (use the London formula) for two He atoms separated by 1.0nm. Relevant data can be found in the Resource section.
-
American Blossom management is planning to make a $3.7 million loan to a French firm. Currently, LIBOR is at 0.55 percent. American management considers a default risk premium of 1.10 percent, a...
-
The following account balances are for Ping Chung, Certified Public Accountant, as of April 30, 20X1. Cash Accounts receivable $120,000 48,000 Maintenance expense 21,400 Advertising expense 17,060...
-
Abbott Equipment leased a protein analyzer to Werner Chemical, Inc., on September 30, 2021. Abbott purchased the machine from NutraLabs, Inc., at a cost of $5 million. The five-year lease agreement...

Study smarter with the SolutionInn App


