At sufficiently high temperatures, the van der Waals equation has the form P L RT>(V m -
Question:
Figure 1.10
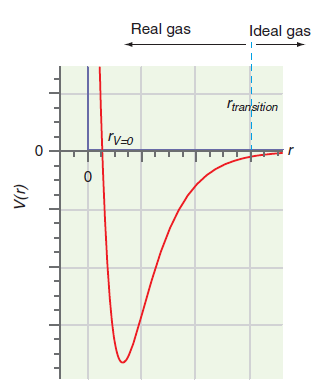
Transcribed Image Text:
Ideal gas Real gas Itrarlsition (1)A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (15 reviews)
At high temperatures the energy of the molecule is lar...View the full answer

Answered By

Zablon Gicharu
I am an educator who possesses the requisite skills and knowledge due to interacting with students for an extended period. I provide solutions to various problems in step-by-step explanations, a well-thought approach and an understandable breakdown. My goal is to impart more straightforward methodologies and understanding to students for more remarkable achievements.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For carbon dioxide gas the constants in the van der Waals equation are = 0.364 J m 3 /mol 2 and (a) If 1.00 mol of gas at 350 K is confined to a volume of find the pressure of the gas using the...
-
In Sample Exercise 10.16, we found that one mole of Cl2 confined to 22.41 L at 0oC deviated slightly from ideal behavior. Calculate the pressure exerted by 1.00 mol Cl2 confined to a smaller volume,...
-
The parameter a in the van der Waals equation is greater for H 2 O than for He. What does this say about the difference in the form of the potential function in Figure 1.10 for the two gases? Figure...
-
A mortgage loan officer uses math on a continual basis during the mortgage lending process. When a prospective borrower applies for a loan, many calculations are made: debt-to-income- ratio,...
-
The following transactions have also occurred at Fitzgerald. 1. Options were granted on July 1, 2013, to purchase 200,000 shares at $15 per share. Although no options were exercised during fiscal...
-
Which of the following retirement plans is eligible for a QDRO? I. 403(b). II. 401(k). III. SEP. IV. SIMPLE IRA. A. I, II. B. II. C. I, III, IV. D. III, IV.
-
Evaluate the performance of each of Alternative Tyress salespeople.
-
Jones Co. is in a technology-intensive industry. Recently, one of its competitors introduced a new product with technology that might render obsolete some of Joness inventory. The accounting staff...
-
To more efficiently manage its inventory, Treynor Corporation maintains its internal inventory records using first-in, first-out (FIFO) under a perpetual inventory system. The following information...
-
1. What important career factors should Brad consider when evaluating his options? 2. What important personal factors should Brad consider when deciding among his career options? 3. What...
-
Define the seven aspects of customer service.
-
What are some uses for hydrogen fuel cells?
-
Henredon can spend \($190,000\) now, similar to the investment in problem 25, for a design portfolio with a different furniture look inspired by some of the ultramodern culture in the metropolitan...
-
What VLANs are allowed, by default, on a trunk port?
-
A zebra can appear highly intelligent or not so intelligent, depending on what?
-
A ( n ) _ _ _ _ _ _ _ _ _ _ is a collection of data organized in a manner that allows access, retrieval, and use of that data. File management system Database management system Integrated system...
-
Problem Management, Change Management, and Incident Management are all key processes of which framework? 1 point ITIL PCI - DSS NIST 8 0 0 - 5 3 GDPR
-
Sales $46,298,115.00 Retained earnings $12,717,254.00 Other expenses $ 5,870,865.00 Notes Payable $ 2,439,553.00 Net PP&E $20,381,945.00 Long-term debt $ 6,300,000.00 Inventory $ 1,235,161.00...
-
Answer each of the following. When appropriate, fill in the blank to correctly complete the sentence. The following nonlinear system has two solutions, one of which is (3, _______). x + y = 7 x 2 + y...
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
For PbI2(s) = 0Pb+(aq) + 2 r(aq), K = 1.4 X 10-8 at 25C and the standard Gibbs energy of formation ofPbI2(s) is -173.64 k] mol ". Calculate the standard Gibbs energy of formation of PbI2 (aq).
-
Write the cell reaction and electrode half-reactions and calculate the standard emf of each the following cells: (a) Ptl C12 (g) I HCl (aq) 11 K, Cr04 (aq) IAg, Cr04(s) IAg (b) Pt 1 Fe3+(aq),Fe2+(aq)...
-
Devise cells in which the following are the reactions and calculate the standard emf in each case: (a) 2 Na(s) + 2 H20 (l) --7 2 NaOH (aq) + H2 (g) (b) H2 (g) + I2 (g) --72 HI (aq) (c) H30+ (aq) + OW...
-
- A two-dimensional vector makes an angle of 0 = 68.3 and has an x-component of 39.1 meters. Calculate the magnitude of this two- dimensional vector. Please enter a numerical answer below. Accepted...
-
a-1.If the required return is 11 percent, what is the profitability index for both projects? (Do not round intermediate calculations and round your answers to 3 decimal places, e.g., 32.161.) Project...
-
Perform the indicated operation (6x313x24x+5)=(x+1)

Study smarter with the SolutionInn App


