Calculate the RedlichKwong parameters of fluorine from the values of the critical constants and compare your results
Question:
Table 7.4
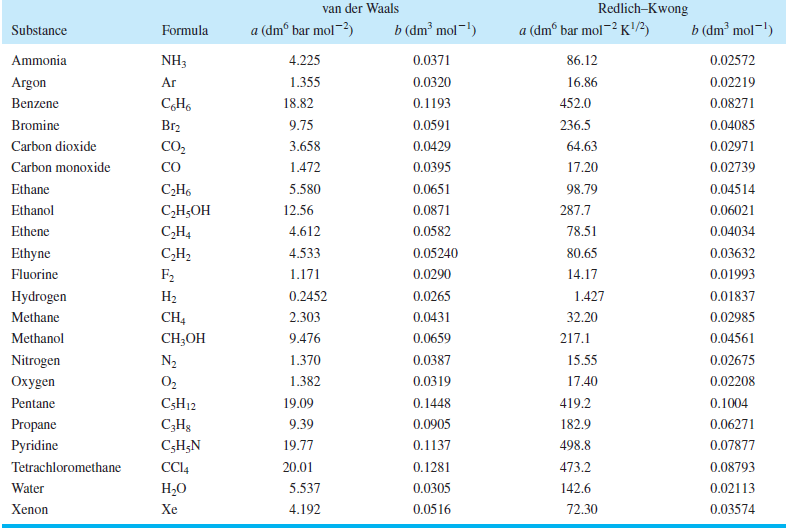
Transcribed Image Text:
Redlich–Kwong van der Waals b (dm² mol¬!) a (dmº bar mol-) b (dm² mol-!) a (dmº bar mol~² K) Substance Formula NH3 0.0371 Ammonia 4.225 86.12 0.02572 Argon Ar 1.355 0.0320 16.86 0.02219 452.0 Benzene 18.82 0.1193 0.08271 Br2 9.75 236.5 Bromine 0.0591 0.04085 CO2 0.0429 Carbon dioxide 3.658 64.63 0.02971 CO Carbon monoxide 1.472 0.0395 17.20 0.02739 0.04514 Ethane С-Н. 5.580 0.0651 98.79 Ethanol С-Н,ОН 12.56 0.0871 287.7 0.06021 Ethene C,H4 4.612 0.0582 78.51 0.04034 CH2 F2 0.05240 0.03632 Ethyne 4.533 80.65 Fluorine 1.171 0.0290 14.17 0.01993 1.427 Hydrogen На 0.2452 0.0265 0.01837 CH4 CH-OН 2.303 0.0431 Methane 32.20 0.02985 0.0659 Methanol 9.476 217.1 0.04561 Nitrogen 0.02675 N2 1.370 0.0387 15.55 0.0319 Охуgen O2 1.382 17.40 0.02208 Pentane C5H12 19.09 0.1448 419.2 0.1004 Propane C;H3 9.39 0.0905 182.9 0.06271 Pyridine C;H;N 19.77 0.1137 498.8 0.07877 Tetrachloromethane CC4 20.01 0.1281 473.2 0.08793 Water Н.О 5.537 0.0305 142.6 0.02113 4.192 Xenon Xe 0.0516 72.30 0.03574
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (13 reviews)
We use the values for the critical consta...View the full answer

Answered By

Saud Ur Rehman
Evaluating manufacturing processes by designing and conducting research programs; applying knowledge of product design, fabrication, assembly, tooling, and materials; conferring with equipment vendors; soliciting observations from operators. Developing manufacturing processes by studying product requirements; researching, designing, modifying, and testing manufacturing methods and equipment; conferring with equipment vendors. Keeping equipment operational by coordinating maintenance and repair services; following manufacturer's instructions and established procedures; requesting special service.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
Calculate the van der Waals parameters of carbon dioxide from the values of the critical constants and compare your results with the values for and b in Table 7.4. Table 7.4 RedlichKwong van der...
-
(a) 537 R (b) 3240 R. Compare your results with the KP values listed in Table A-28. TABLE A-28 Natural logarithms of the equilibrium constant K The equilibrium constant kp for the reaction AA+cC+D is...
-
(a) 298 K (b) 1800 K. Compare your results with the KP values listed in Table A-28. TABLE A-28 Natural logarithms of the equilibrium constant K The equilibrium constant kp for the reaction AA+cC+D is...
-
As a brand manager, for a product of your choice, develop a strategy for launching the product in a manner most likely to capture the attention of the product's primary target market. Write a...
-
Describe at least three things that your organization is doing to encourage ethical behavior and corporate social responsibility and at least three things that you think it should be doing to...
-
Who benefits and loses from brain drain?
-
The date of a management representation letter coincides with the: a. Date of the auditor's report. b. Balance sheet date. c. Date of the latest subsequent event referred to in notes to the financial...
-
The records of Alaska Company provide the following information for the year ended December 31. Required 1. Use the retail inventory method to estimate the companys year- end inventory at cost. 2. A...
-
A company sold 5,600 units of its only product and reported the following contribution margin income statement for this year. Contribution Margin Income Statement For Year Ended December 31 Sales...
-
Following high school, James Sinegal worked for discount warehouse Fed Mart unloading mattresses. Then,in the 1970s as an employee of California-based PriceClub, he absorbed every detail of the...
-
Which is NOT a proper way to express the ratio of 10 out of each 100? a. 10 b. 0.10 c.10% d. 10/100
-
Have traditional patterns of consumption been radically changed by globalization?
-
Aaron contributed $2000 to his RRSP at the end of every half-year. What was the value of his RRSP after 12 1/2 years if the RRSP grew at 7.5% compounded semiannually?
-
3. Assets A and B are perfectly negatively correlated and have expected returns and standard deviations as follows: Asset A B Expected Return 10 Standard Deviation 8 16 11 What would be the min. you...
-
Growth and Financing Mr. Miyagi is very excited because sales for his nursery and plant company are expected to double from $600,000 to $1,200,000 next year. Mr. Miyagi notes that net assets (Assets...
-
A homeowner is using a personal loan to borrow $18,000 to replace the roof. The loan offers the option of making no payments for the first 12 months, during which the interest is compounded monthly...
-
Hedging with Forwards and Options: One year from today, you will be receiving CHF 500,000, but you would prefer dollars. The current spot rate is CHF 0.92 per USD, and the one year forward rate is...
-
A corporate bond has an annual coupon rate of 9%, payable semiannually, and a maturity of 25 years. Determine the bond's yield to maturity if its current yield is 7%.
-
Determine whether each integral is convergent or divergent. Evaluate those that are convergent. 1 = dx V1 + x
-
What is the amount of total interest dollars earned on a $5,000 deposit earning 6% for 20 years?
-
Prove the following fact about power series: If two power series in the same independent variable are equal to each other for all values of the independent variable, then any coefficient in one...
-
Find the interval of convergence for the series for cos (x).
-
Find the interval of convergence for the series for sin (x).
-
9 1. Compute the indefinite integral. +1 (3+ 4+4) dr.
-
Sarah Jackson Antiques issued its 11%, 10-year bonds payable at a price of $338,220 (face value is $400,000). The company uses the straight-line amortization method for the bond discount or premium....
-
Suppose you start with a full tank of gas (14 gallons) in your truck. After driving 6 hours, you now have 6 gallons left. If x is the number of hours you have been driving, then y is the number of...

Study smarter with the SolutionInn App


