Calculate the standard enthalpies of formation of (a) KClO 3 (s) from the enthalpy of formation of
Question:
Calculate the standard enthalpies of formation of
(a) KClO3(s) from the enthalpy of formation of KCl,
(b) NaHCO3(s) from the enthalpies of formation of CO2 and NaOH together with the following information:
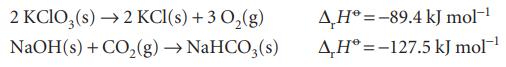
Transcribed Image Text:
2 KClO3(s) → 2 KCl(s) + 3 O₂(g) NaOH(s) + CO₂(g) → NaHCO3(s) AH-89.4 kJ mol-¹ AH-127.5 kJ mol-¹
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (10 reviews)
Solution a From the data given enthalpy of formation of ClO3 is 9626 kJ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The standard enthalpies of formation of gaseous propyne (C3H4), propylene (C3H6), and propane (C3H8) are + 185.4, +20.4, and -103.8 kJ/mol, respectively. (a) Calculate the heat evolved per mole on...
-
`The standard enthalpies of formation of ions in aqueous solutions are obtained by arbitrarily assigning a value of zero to H+ ions; that is, Hf [H+(aq)] = 0. (a) For the following reaction ...
-
The standard enthalpies of formation of S(g), F(g), SF4(g), and SF6(g) are 1278.8 kJ/ mol, 179.0 kJ/ mol, 775 kJ/ mol, and 1209 kJ/ mol, respectively. a. Use these data to estimate the energy of an...
-
What is a personal balance sheet? Name three classifications of assets. Briefly define and give examples of each.
-
The City of Columbia is trying to attract a new manufacturing business to the area. It has offered to install and operate a water pumping plant to provide service to the proposed plant site. This...
-
Assume that Cycle Guys bought and sold a line of mountain bikes during December as follows: Cycle Guys uses the perpetual inventory system. Requirements 1. Compute the cost of ending inventory under...
-
Prince Corporation holds 75 percent of the common stock of Sword Distributors Inc., purchased on December 31, 20X1, for $2,220,000. At the date of acquisition, Sword reported common stock with a par...
-
Howell Corporation has a job order cost system. The following debits (credits) appeared in Work in Process for the month of July: July 1, balance....... July 31, direct materials.. July 31, direct...
-
It is false that no A are B. Therefore, some A are B. Use the modified Venn diagram technique to determine if the following immediate inference forms are valid from the Boolean standpoint,...
-
Show that the following functions have exact differentials: (a) X 2 y + 3y 2 , (b) X cos xy, (c) X 3 y 2 , (d) T(t + e s ) + s.
-
Calculate the standard enthalpy of solution of AgCl(s) in water from the enthalpies of formation of the solid and the aqueous ions.
-
In a Gallup Poll conducted October 2010,44% of the people polled answered more strict to the following question:Do you feel that the laws covering the sale of rearms should be made more strict, less...
-
How does lean production improve lead times and customer responsiveness?
-
How does lean production impact quality management and reduce defects in the manufacturing process?
-
a) Distinguish between duality theorems I and II. [6 Marks] b) Using a two-product two constraint case for a firm production characterise and discuss the duality of the maximization problem. [5...
-
How does lean production incorporate the concept of "pull" to synchronize production with customer demand?
-
A string is wound around a uni radius 0.4 m and mass 3 kg. The disc is released from rest with the string vertical and its top end tied to a fixed support. h F: 3 kg W 0.4 m Calculate the speed of...
-
The functional use test is often used to determine whether the replacement property is similar or related in service or use to the property converted. Explain the functional use test.
-
Find the work done in pumping all the oil (density S = 50 pounds per cubic foot) over the edge of a cylindrical tank that stands on one of its bases. Assume that the radius of the base is 4 feet, the...
-
What are the advantages of working with normalized wavefunctions?
-
Functions of the form sin(nx/L) can be used to model the wavefunctions of electrons in a carbon nanotube of length L. Show that the wavefunctions sin(nx/L) and sin(mx/L), where nm, are orthogonal for...
-
A particle free to move along one dimension x (with 0x
-
Question 3 (4 marks) Students at the school who are studying mathematics in Years 7 to 10 can receive one of three grades at the end of the year: distinction (D), credit (C) or pass (P). A regular...
-
How has technology brought about social change in your lives in comparison to your parents' and your grandparents' generation? how has technology brought about change in your grandparents'/parents'...
-
Identify the role and function of the major components of the Canadian criminal justice system Demonstrate the manner in which the criminal justice system components relate and interact with each...

Study smarter with the SolutionInn App


