Can the individual states in Table 22.1 be distinguished experimentally? States and Terms for the np? Configuration
Question:
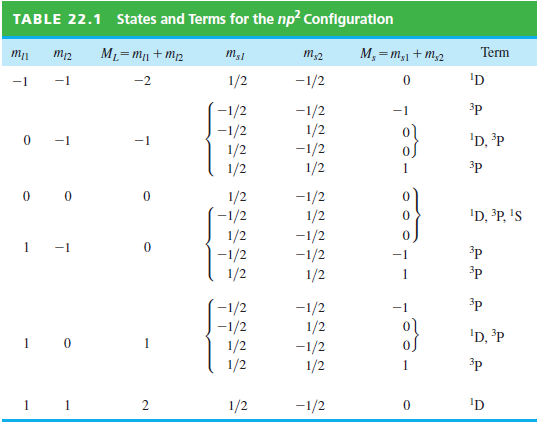
Transcribed Image Text:
States and Terms for the np? Configuration TABLE 22.1 M, =m,1 + m,2 Term ML=mn + M2 m;1 m,2 'D -2 1/2 -1/2 -1 3p -1/2 -1/2 1/2 -1/2 1/2 -1 -1/2 1/2 1/2 'D, ³P -1 -1 3p 1/2 -1/2 -1/2 1/2 'D, ³P, 's 1/2 -1/2 1/2 -1/2 1 -1 -1/2 1/2 ³p 1 3p -1/2 -1/2 1/2 -1/2 1/2 1/2 'D, ³P -1/2 1/2 3p 'D 2 1/2 -1/2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
No The energy level that corresponds to an individual ...View the full answer

Answered By

Payal Mittal
I specialize in finance and accounts.You can ask any question related to til undergradution.Organizational behaviour and HRM are my favourites for you can always relate to them and is an art with practical knowledge base.
4.90+
226+ Reviews
778+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How might continuous and discontinuous modes of DNA replication be distinguished experimentally?
-
Table 8.1 shows that Turner syndrome occurs when an individual inherits one X chromosome but lacks a second sex chromosome. Can Turner syndrome be due to nondisjunction during oogenesis,...
-
The technique of Western blotting can be used to detect proteins that are translated from a particular mRNA. This method is described in Chapter 20 and also in experimental question E5. Let's suppose...
-
Which of these is not a feature of the AES encryption cipher? Question 4 1 4 1 Answer a . . Its fast execution time. b . . Its use of Feistal networks. c . . Its ease of use in common programming...
-
Find the area of the sector of a circle of radius r and central angle . Radius........................... Central Angle 1. 20 inches........................... 1500 2. 7.5 millimeters...
-
Can you name five types of channels of communication?
-
Identify and discuss the alternative courses of action for an auditor when his sample results indicate that a control cannot be relied upon.
-
Donahue Company uses both special journals and a general journal as described in this chapter. On June 30, after all monthly postings had been completed, the Accounts Receivable control account in...
-
You wish to purchase a new house 20 years from today. You currently have $25,000 debt you need to pay off in year 4. Interest rate is 15%. You can also save $3,500 a year. What kind of a house can...
-
In filling bags of nitrogen fertilizer, it is desired to hold the average overfill to as low a value as possible. The lower specification limit is 22.00 kg (48.50 lb), the population means weight of...
-
How is it possible to determine the L and S values of a term knowing only the M L and M S values of the states?
-
Write an equation giving the relationship between the Rydberg constant for H and for Li 2+ .
-
Pembroke Company had poor internal control over its cash transactions. The following are facts about its cash position at November 30: The cash books showed a balance of $18,901.62, which included...
-
Analyze the "National Models of Healthcare Delivery". Develop your ideal model for healthcare delivery in the United States.
-
Conduct a General Market Potential (covering market size, market growth, and quality of demand) and Site Potential of the chosen country.
-
Sales of personal care products tend to remain stable during recessionary periods. Classify the industry sector of personal care products.
-
Sheridan Construction Company has entered into a contract beginning January 1, 2025, to build a parking complex. It has been estimated that the complex will cost $596,000 and will take 3 years to...
-
Discuss emerging trends and advancements in IPC research, such as lock-free synchronization mechanisms, zero-copy data transfers, and techniques for minimizing latency in inter-process communication .
-
Determine whether each infinite geometric series converges or diverges. If it converges, find its sum. 1 + 3
-
The Home Depot is the leading retailer in the home improvement industry and one of the 10largest retailers in the United States. The company included the following on its January 29, 2012, balance...
-
The densities of air at 85C, 0C, and 100C are 1.877 g dm 3 , 1.294 g dm 3 , and 0.946 g dm 3 , respectively. From these data, and assuming that air obeys Charless law, determine a value for the...
-
The density of a gaseous compound was found to be 1.23 kg m 3 at 330 K and 20 kPa. What is the molar mass of the compound?
-
The second virial coefficient of methane can be approximated by the empirical equation B(T) = a + be c/T2 , where a = 0.1993 bar 1 , b = 0.2002 bar 1 , and c = 1131 K 2 with 300 K < T < 600 K. What...
-
Some enterprising physics students working on a catapult decide to have a water balloon fight in the school hallway. The ceiling is of height 3 . 4 m, and the balloons are launched at a velocity of 9...
-
1-Define electric fields and how it helps us understand electricity. 2-Electric fields are represented as a physical effect of a configuration of charges that is created by the attraction of electric...
-
Why thin film on a glass surface can remove completely reflections at certain light wavelength? Magnesium fluoride (n = 1.38) is used as coating on nonreflecting glass. How thin coating is enough to...

Study smarter with the SolutionInn App


