Consider the arrangement shown in 5 for a system consisting of an O-H group and an O
Question:
Consider the arrangement shown in 5 for a system consisting of an O-H group and an O atom, and then use the electrostatic model of the hydrogen bond to calculate the dependence of the molar potential energy of interaction on the angle θ.
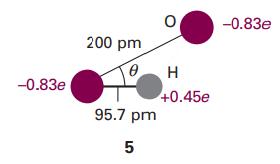
Transcribed Image Text:
-0.83e 200 pm 0 95.7 pm 5 H +0.45e -0.83e
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
The potential energy of interaction between two molecules is given by Uq1q24...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Consider the arrangement shown in Figure P31.20. Assume that R = 6.00 Ω, = 1.20 m, and a uniform 2.50-T magnetic field is directed into the page. At what speed should the bar be moved to...
-
Consider the arrangement shown in figure. Assume that R = 6.00 and = 1.20 m, and that a uniform 2.50-T magnetic field is directed into the page. At what speed should the bar be moved to produce a...
-
Consider a system consisting of an ice cube. (a) Under what conditions can the ice cube melt reversibly? (b) If the ice cube melts reversibly, E is zero for the process? Explain.
-
Approximately 8.33% of men are colorblind. You survey men from a large population until you find one who is colorblind. a. Explain in words what X is in this situation and what values it can take. b....
-
Describe the three-tier client-server model.
-
Explain what is meant by the following statement. Please include proper citations in your discussion post The performance report does not compare actual performance to the flexible budget. Therefore,...
-
How do you forecast the Problem Spaces?
-
A firm is faced with the attractive situation in which it can obtain immediate delivery of an item it stocks for retail sale. The firm has therefore not bothered to order the item in any systematic...
-
Talk Technologies manufactures capacitors for cellular base stations and other communications applications. The company's July 2022 static budget was based on expected sales of 12,000 units is...
-
Fort Worth, Inc., specializes in manufacturing some basic parts for sports utility vehicles SUVs) that are produced and sold in the United States. Its main advantage in the United States is that its...
-
Suppose you distrusted the Lennard-Jones (12,6) potential for assessing a particular polypeptide conformation, and replaced the repulsive term by an exponential function of the form e r r/0 . (a)...
-
The magnitude of the electric field at a distance r from a point charge Q is equal to Q/4 0 r 2 . How close to a water molecule (of polarizability volume 1.4810 30 m 3 ) must a proton approach before...
-
Burke Copy Center purchased a machine on January 1, 2012, for $180,000 and estimated its useful life and salvage value at ten years and $30,000, respectively. On January 1, 2017, the company added...
-
Durham Limited had an authorised capital of 200,000 divided into 100,000 ordinary shares of 1 each and 200,000 8 per cent preference shares of 50p each. The following balances remained in the...
-
Grant and Herd are in partnership sharing profits and losses in the ratio 3 to 2. The following information relates to the year to 31 December 2015: 1 The partnership agreement allows for Herd to be...
-
Upon graduation, Jeffrey Feldhusen borrows $15,000 to finance a late model used car. The loan is made by a family member who wishes to have equal annual payments at 9 percent over 4 years. a. How...
-
The file HybridSales contains the number of domestic and imported hybrid vehicles sold in the United States from 1999 to 2016. You want to be able to predict the number of domestic and imported...
-
Draw a graph of the Lee familys supply of loanable funds curve. In 2020, the Lee family had disposable income of $80,000, wealth of $140,000, and an expected future income of $80,000 a year. At a...
-
How many grams of KOH are present in 35.0 mL of a 5.50 M solution?
-
Calculate the speed that a gas-phase fluorine molecule would have if it had the same energy as an infrared photon ( = 1.00 10 4 nm), a visible photon ( = 500. nm), an ultraviolet photon ( = 100....
-
Calculate the highest possible energy of a photon that can be observed in the emission spectrum of H.
-
What is the maximum number of electrons that can be emitted if a potassium surface of work function 2.40 eV absorbs 5.00 10 3 J of radiation at a wavelength of 325 nm? What is the kinetic energy and...
-
What is the balance in Work in Process Inventory at the end of each month? Work in Process Inventory April 30 $ May 31 A 10,140 15,950 June 30 $ 10,450
-
Sako Company s Audio Division produces a speaker used by manufacturers of various audio products. Sales and cost data on the speaker follow: Selling price per unit on the intermediate market $ 6 0...
-
Vulcan Company's contribution format income statement for June is as follows: Vulcan Company Income Statement For the Month Ended June 30 Sales Variable expenses Contribution margin Fixed expenses...

Study smarter with the SolutionInn App


