Consider the equilibrium CO(g) + H2O(g) ???? CO2(g) + H2(g). At 1150. K, the composition of the
Question:
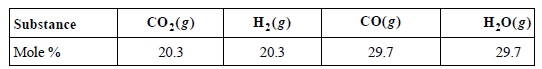
a. Calculate KP and ΔGoR at 1150. K.
b. Given the answer to part (a), use the ΔHof of the reaction species to calculate ΔGoR at 298.15 K. Assume that ΔHR° is independent of temperature.
Transcribed Image Text:
CO(g) co:(g) H;(g) H,0(g) Substance 20.3 29.7 Mole % 20.3 29.7
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
a COg H 2 OgCO 2 g H 2 g For each compo...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A chamber initially contains a gaseous mixture consisting of 4 kmol of CO2, 8 kmol of CO and 2 kmol of H2. Assume an equilibrium mixture formed consists of CO2, CO, H2O, H2 and O2 at 2600 K and 100...
-
At 25 C, Kp < 1 10-31 for the reaction N2(g) + O2(g) 2NO(g) a. Calculate the concentration of NO (in molecules/ cm3) that can exist in equilibrium in air at 25oC. In air PN2 = 0.8 atm and PO2 = 0.2...
-
Consider the water-gas-shift reaction: H2(g) + CO2(g) H2O(g) + CO(g) At high temperatures and low to moderate pressures the reacting species form an ideal- gas mixture. By Eq. (11.27): When the Gibbs...
-
Show that plane stress displacements for the Flamant problem in Section 8.4.7 under only tangential force X are given by: Data from section 8.4.7 My (1 v) - - -0 sin (1 v)X, -0 cost + 2X, 20 log r...
-
List and explain the stages in the product life cycle. How can a small firm extend its product's life?
-
Which costing system is most likely to produce the least cost distortion? a. Traditional costing system b. Plantwide overhead rate c. Departmental overhead allocation rates d. Activity-based costing
-
Explain the purpose of the temperature variable in simulated annealing. How effective would the method be without it?
-
Gamma Corporation is considering the installation of governors on cars driven by its sales staff. These devices would limit the car speeds to a preset level, which is expected to improve fuel...
-
Your client, Mr Imtiaz Khan is assessing two different types of 4-year mutually exclusive investments. From his analysis, he anticipates generating future end-of-year cash flows arising from each of...
-
Which of the graphs in Fig. Q25.12 best illustrates the current I in a real resistor as a function of the potential difference V across it? Explain. Figure Q25.12 (a) (b) (c) (d)
-
Calculate K P at 600.K for the reaction N 2 O 4 (l) 2NO 2 (g) assuming that H o R is constant over the interval 298 725 K.
-
When 2,4-dibromo-3-methyltoluene is treated with bromine in the presence of iron (Fe), a compound with molecular formula C8H7Br3 is obtained. Identify the structure of this product.
-
The function D defined by D(x) = 2375x 2 + 5134x + 5020 models AIDS deaths x years after 1984. Write a formula g(x) that computes AIDS deaths during year .x, where x is the actual year.
-
1. What are some strategies or techniques you use to move through the different stages of the creative process? 2. In your experience, which stage of the creative process do you find to be the most...
-
A brand new machine is purchased at the beginning of the year for $620,000. Its useful life is estimated at 6 years with a salvage value estimated at $100,,000. Using the straight line method, how...
-
You have been approached by a local news reporter asking if it would be beneficial or not to have a mobile integrated health (MIH) or community paramedic (CP) program aimed at assisting the elderly...
-
What are some of the most common feelings that you have in your day-to-day experience at school? Where do you feel this feeling in your body? Where else have you felt this way?
-
Shelby's retirement fund has an accumulated amount of $55,000. If it has been earning interest at 2.13% compounded monthly for the past 25 years, calculate the size of the equal payments that she...
-
Find the area of the finite part of the paraboloid y = x 2 + z 2 cut off by the plane y = 25.
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
Find the real root of the equation x 2 e x = 0.
-
Find the root of the equation x 2.00 sin (x) = 0.
-
Find two positive roots of the equation ln (x) 0.200x = 0.
-
Excerpts from Andre Company's December 31, 2024 and 2023, financial statements are presented below: Accounts receivable Inventory Net sales Cost of goods sold Total assets Net income Total...
-
The York City Hospital has just acquired new equipment. The equipment cost $ 4 , 2 5 0 , 0 0 0 , and the organization spent $ 1 3 5 , 0 0 0 on upgrading the physical plant to the new equipment will...
-
Calculate the following showing all the necessary steps: Note: CPP rate to be used in the calculation is 5.95%. Note: Don't forget to deduct pay period exemption\ \ Tanya earns $25.00 per hour. This...

Study smarter with the SolutionInn App


