Consider the following mechanism for ozone thermal decomposition: a. Derive the rate law expression for the loss
Question:
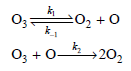
a. Derive the rate law expression for the loss of O3(g).
b. Under what conditions will the rate law expression for O3(g) decomposition be first order with respect to O3(g)?
Transcribed Image Text:
:02 +0 O3 03 +0- 202
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
a b Atomic oxygen is the intermediate species and the diffe...View the full answer

Answered By

Muhammad Mahtab
everyone looks that their work be perfect. I have more than a five year experience as a lecture in reputable institution, national and international. I provide perfect solution in marketing, case study, finance problems, blog writing, article writing, business plans, strategic management, human resource, operation management, power point presentation and lot of clients need. Here is right mentor who help clients in their multi-disciplinary needs.
5.00+
3+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following mechanism for renaturation of a double helix from its strands A and B: Derive the rate equation for the formation of the double helix and express the rate constant of the...
-
Consider the following mechanism for the thermal decomposition ofR2: Where R2, PA' PB are stable hydrocarbons and Rand R' are radicals. Find the dependence of the rate of decomposition of R, on the...
-
Consider the following mechanism for a reaction in aqueous solution and indicate the species acting as a catalyst: Explain why you believe this species is a catalyst. What is the overall reaction?...
-
Which of the following statements is correct regarding a work sheet and the adjustment process? Adjusting journal entries are prepared from the Adjusted Trial Balance columns. Adjusting journal...
-
Find the inverse function of f informally. Verify that f (f-1 (x)) = x and f-1(f (x)) = x. 1. f(x) = 6x 2. f(x) = 1/3x 3. f(x) = 3x + 1
-
When should you engage an attorney? What criteria will you use in your decision?
-
"For a given chemical process, the net heat change will be the same whether the process occurs in one or in several stages." This is known as (a) Kirchoff's law (b) Hess's law (c) Laplace's law (d)...
-
Merton Manufacturing Company has an opportunity to purchase some technologically advanced equipment that will reduce the companys cash outflow for operating expenses by$1,280,000 per year. The cost...
-
Bloomtech Ltd (Bloomtech) is one of the top software development companies in australia. However recently it has lost a number of its key clients to its competitors and consequently ha sbeen having...
-
Wolfgang Iser, the accountant of Starkuchen, wants to further examine the relative profitability of raisin cake and layered carrot cake. He questions the accuracy of the activity-based normal costing...
-
Consider the formation of double-stranded (DS) DNA from two complementary single strands (S and S²) through the following mechanism involving an intermediate helix (IH): a. Derive the rate law...
-
Determine the expression for fractional coverage as a function of pressure for the dissociative adsorption mechanism described in the text in which adsorption is accompanied by dissociation: R,(g) +...
-
Refer to the Crystal Springs Bottling Department Data Set and your answers to S5- 19 and S5- 20. Complete Step 5 of the process costing procedure: Assign costs to units completed and to units in...
-
If you have a car with TPMS, it monitors the pressure in your tires continuously. As you were driving to class on January 11, the temperature of the air in your tires got up to 45 C from friction...
-
A microwave oven operates at 2.5 GHz. Assume that 5% of microwave photons are absorbed by 200 ml of water kept inside the oven. The time needed to warm the water from 20Cto 70C is 2 minute. Specific...
-
An insulating sphere is 7 . 4 3 cm in diameter and carries a 5 . 0 5 uC charge uniformly distributed throughout its interior volume. Calculate the charge enclosed by a concentric spherical surface...
-
While training on Earth, an astronaut was able to jump 5 6 . 1 cm above ground, as measured by the maximum height reached by their feet. Assuming they can jump with the same initial velocity...
-
Solve the inequality. x+2x-630
-
On a given day, the American Baseball League schedules 7 games. How many different outcomes are possible, assuming that each game is played to completion?
-
1. What is the semi-annually compounded interest rate if $200 accumulates to $318.77 in eight years? Answer in percentage with two decimal places. 2. What is the quarterly compounded interest rate if...
-
Predict the products for each of the following reactions: a. b. c. d. . Ti[OCH(CH,),1. (+)-DET -- THCICH) (-)-DET
-
Benzoic acid, 1.35 g, is reacted with oxygen in a constant volume calorimeter to form H 2 O(l) and CO 2 (g) at 298 K. The mass of the water in the inner bath is 1.55 10 3 g. The temperature of the...
-
Calculate the P and T values for which Br2(g) is in a corresponding state to Xe(g) at 330. K and 72.0 bar.
-
X1155: Throwing an Exception The method below takes in a String parameter representing a username. It should throw an Exception if the username is less than 3 characters long. The method correctly...
-
Catch Exception The method divideValues() will throw an IllegalArgumentException if either parameter is 5. The method below takes two integer values as parameters and calls divideValues() in a try...
-
Wyoming Products Inc. completed and transferred 145,000 particle board units of production from the Pressing Department. There was no beginning inventory in process in the department. The ending...

Study smarter with the SolutionInn App


