Consider the phase diagram in Fig. 6.44, which represents a solidliquid equilibrium. Label all regions of the
Question:
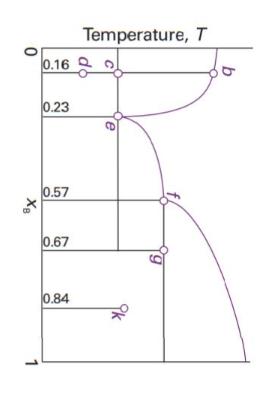
Consider the phase diagram in Fig. 6.44, which represents a solid–liquid equilibrium. Label all regions of the diagram according to the chemical species that exist in that region and their phases. Indicate the number of species and phases present at the points labelled b, d, e, f, g, and k. Sketch cooling curves for compositions xB = 0.16, 0.23, 0.57, 0.67, and 0.84.
Fig. 6.44
Transcribed Image Text:
0 XB 0.16 0.23 0.57 0.67 0.84 Temperature, T b
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
answer The number of species and phases present at each labeled point are Poin...View the full answer

Answered By

Ma Kristhia Mae Fuerte
I have extensive tutoring experience, having worked as a private tutor for over three years. I have tutored students from different academic levels, including high school, undergraduate, and graduate levels. My tutoring experience has taught me to be patient, attentive to student needs, and effective in communicating difficult concepts in simple terms.
I have a strong background in statistics, probability theory, data analysis, and data visualization. I am proficient in using statistical software such as R, Python, and SPSS, which are commonly used in academic research and data analysis. Additionally, I have excellent communication and interpersonal skills, which enable me to establish rapport with students, understand their learning styles, and adapt my teaching approach to meet their needs.
I am passionate about teaching and helping students achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the phase diagram in Fig. 5.6, which represents a solidliquid equilibrium. Label all regions of the diagram according to the chemical species that exist in that region and their phases....
-
The phase diagram in figure can be interpreted to yield information on how the boiling and melting points of water change with altitude. (a) Explain how this information can be obtained. (b) How...
-
A B C D E F G 1 Suppose the risk free interest rate is 4%. Calculate the following using the EXCEL functions. 2 3 Having $500 today is equivalent to having what amount in one year? 4 Present value...
-
Because Natalie has been so successful operating Cookie Creations, Katy would like to have Natalie become her partner. Katy believes that together they will create a thriving cookie-making business....
-
A forward market is trading for future purchase of a commodity, while a spot market is trading for immediate purchase. Is the stock market a spot or forward market?
-
Two moles of an ideal gas are compressed in a cylinder at a constant temperature of 85.0C until the original pressure has tripled. (a) Sketch a pV-diagram for this process. (b) Calculate the amount...
-
It is generally known that boys grow at an unusually fast rate between the ages of about 12 and 14. Following are heights, in inches, of 40 boys measured at age 12 and again at age 14. Can you...
-
Kraft Foods successfully introduced DiGiorno Pizza into the marketplace in 1996, with first year sales of $120 million, followed by $200 million in sales in 1997. It was neither luck nor coincidence...
-
Piper Products sold 426,000 units during the last period when industry volume totaled 3.00 million units. The company originally expected to sell 462,500 based on a budgeted market share of 15...
-
Creating a Partnership Tax Return Overview You are a senior tax accountant in the firm, Ernest & Rainhouse. Joe, a new junior accountant, has just completed an interview with a new client Harry,...
-
State the number of components in the following systems. (a) NaH 2 PO 4 in water at equilibrium with water vapour but disregarding the fact that the salt is ionized. (b) The same, but taking into...
-
Phosphorus and sulfur form a series of binary compounds. The best characterized are P 4 S 3 , P 4 S 7 , and P 4 S 10 , all of which melt congruently. Assuming that only these three binary compounds...
-
Employees are paid every Saturday for the preceding work week. If a balance sheet is prepared on Wednesday, December 31, what does the amount of wages earned during the first three days of the week...
-
= Hornes is admitted to the partnership of Rose & Novak. Prior to Hornes' admission, the partnership books show Rose's capital balance at $170,000 and Novak's at $85,000. Assume Rose and Novak share...
-
From the units' readings, we note that the chances of risk events occurring and their respective costs increasing change over the project life cycle. Assume you are a project manager of a...
-
Faucet Industries Inc. manufactures faucets in a small manufacturing facility. The faucets are made from zinc. Faucet Industries has 60 employees. Each employee presently provides 35 hours of labor...
-
A rock is thrown from a cliff top at 18 m/s, 25 degrees above the horizontal. It lands on the beach 4.2 seconds later. a) What is the height of the cliff? b) How far from the base of the cliff did...
-
01. Find the accumulated amount. P 1,000 5,250 7,235 10,500 300 02. Find the accumulated amount. P 3,500 3,750 1,875 12,500 250 i (simple interest) 0.5% 5% 2.5% 7% 10% i (simple interest) 0.10% 4%...
-
What seem to be the essential elements of this successful recovery from failure?
-
A 6-lb shell moving with a velocity ?? v0k explodes at point D into three fragments which hit the vertical wall at the points indicated. Fragments A, B, and C hit the wall 0.010 s, 0.018 s, and 0.012...
-
Calculate the ratio A/B for transitions with the following characteristics: (i) 70.8pm X-rays, (ii) 500nm visible light, (iii) 3000 cm 1 infrared radiation.
-
Describe the physical origins of the gross selection rules for infrared spectroscopy.
-
What is the physical interpretation of a selection rule?
-
6 XTL Company manufactures tables and has two divisions, the Production Division and the Assembly Division. The Production Division manufactures tables for the Assembly Division, which completes the...
-
UMPI Corporation manufactures and sells mini robots, a seasonal product that has peak sales in the 3rd quarter. The following information is related to the operations for Year 2 (the upcoming year)...
-
Please insert answer in the highlighted area. Please also include the cell formula in the explanation for better understanding. TranscribedText: Start 1 IN 5 6 7 8 TV Key Assumptions Construction...

Study smarter with the SolutionInn App


