Cyclopropane isomerizes into propene when heated to 500C in the gas phase. The extent of conversion for
Question:
Cyclopropane isomerizes into propene when heated to 500°C in the gas phase. The extent of conversion for various initial pressures has been followed by gas chromatography by allowing the reaction to proceed for a time with various initial pressures:
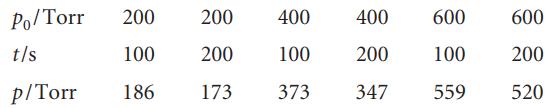
where p0 is the initial pressure and p is the final pressure of cyclopropane.
What are the order and rate constant for the reaction under these conditions?
Transcribed Image Text:
Po/Torr 200 200 t/s 100 200 p/Torr 186 173 400 100 373 400 200 347 600 600 100 200 559 520
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
R k Ax and d Adt Rate for 1st condition Po P t 014 fo...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The decomposition of iodoethane in the gas phase proceeds according to the following equation: C2H5I(g) C2H4(g) + HI(g) At 660. K, k = 7.2 10-4 s-1; at 720. K, k = 1.7 10-2 s-1. What is the rate...
-
The decomposition of acetaldehyde was studied in the gas phase at 791 K. The results of the measurements are shown as follows: Initial concentration (in mol/L) 9.72x10 -3 4.56x10 -3 Half-life (in s)...
-
In the gas phase at 500.C, cyclopropane reacts to form propene in a first-order reaction. The figure below shows the concentration of cyclopropane plotted versus time. Use the graph to calculate...
-
Plot the six risks on a probability/impact matrix using the project impact matrix 2023 template. Write one to two sentences stating the rationale for how you determined the quadrant placement for...
-
Simmons Market Research conducted a national consumer study of 13,787 respondents. A subset of the respondents was asked to indicate the primary source of the vitamins or mineral supplements they...
-
An important parameter in the design of gas absorbers is the ratio of the flow rate of the feed liquid to that of the feed gas. The lower the value of this ratio, the lower the cost of the solvent...
-
9 P 5 Perform the indicated calculation.
-
The City Commission of Nashville has decided to build a botanical garden and picnic area in the heart of the city for the recreation of its citizens. The precedence table for all the activities...
-
Answer the following questions regarding a new California state Law, https://leginfo.legislature.ca.gov/faces/billNavClient.xhtml?bill_id=202120220AB89...
-
Compute the missing amounts In the separate Income statements A, B, and C. Sales 24 21,200 92,000 Cost of goods sold 40,800 30,800 Gross profit 36,200 12,300 Expenses 6,400 Net income 13,800 21,800...
-
A second-order reaction of the type A + B P was carried out in a solution that was initially 0.050 mol dm 3 in A and 0.080 mol dm 3 in B. After 1.0 h the concentration of A had fallen to 0.020 mol...
-
The rate constant for the first-order decomposition of N 2 O 5 in the reaction 2 N 2 O 5 (g) 4 NO 2 (g) + O 2 (g) is k = 3.38 10 5 s 1 at 25C. What is the half-life of N 2 O 5 ? What will be the...
-
How is the concept of a business combination related to the concept of a parent-subsidiary relationship?
-
What happens if a client has funds in an account that exceeds SIPC limits and the broker-dealer becomes insolvent? Explain.
-
Cameron Electronics makes and sells power banks for phones. Each power bank has a selling price of $90. The following cost data per power bank which is based on a capacity of 4,000 units per period....
-
Ben has accumulated a lot of credit card debt over the years, but somehow he always manages to qualify for more credit. He has opened several credit cards that offer an introductory zero percent...
-
Complete the box below to match your ideal values and culture to trends that you and your team can personally engage with to manifest the culture for a new startup. Worth How value becomes an ideal...
-
Lululemon is considering a new project that improves inventory management for the next 5 years. The project has similar risks as the overall firm. The new technology will require an investment in...
-
Why doesn't cholera toxin cause cancer?
-
On July 1, 2011, Flashlight Corporation sold equipment it had recently purchased to an unaffiliated company for $480,000. The equipment had a book value on Flashlights books of $390,000 and a...
-
The rate constant for the decomposition of a certain substance is 1.70 x 10-2 dm3 mol-1 S-1 at 24C and 2.01 X 10-2 dm3 mol-1 S-1 at 37"C. Evaluate the Arrhenius parameters of the reaction.
-
Predict the order of magnitude of the isotope effect on the relative rates of displacement of (a) IH and 3H, (b) 160 and ISO. Will raising the temperature enhance the difference? Take kr(C-H) = 450 N...
-
The effective rate constant for a gaseous reaction that has a Lindemann-Hinshelwood mechanism is 1.7 X 10-3 s-I at 1.09 kPa and 2.2 X 10-4 S-1 at 25 Pa. Calculate the rate constant for the activation...
-
When you visit a retail outlet it has amazing design and space management. Discuss two such retail organisations and their designs one from UAE and another from any other part of the world. Make use...
-
Companies segment their consumer markets based on many different characteristics. For each of the following segmentation variables, give an example of a brand that appears to use that variable to...
-
Q3. Delicious House sells eftali Kebap. In a year (A year has to be considered as 360 days), we sell 18,000 kebaps to our customers. The additional data can be given as follows: Fixed Cost per year:...

Study smarter with the SolutionInn App


