Describe what you would observe if you heated the liquid mixture at the composition corresponding to point
Question:
Figure 9.24b
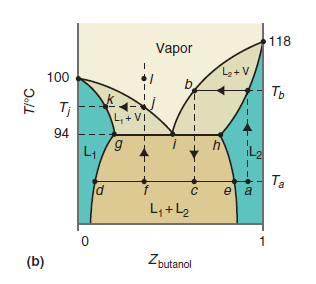
Transcribed Image Text:
118 Vapor 100 Ть Tj L + VI 94 6, L1 Ta e a L,+L2 Zbutanol (b) T/°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The mixture of two liquids will increase in temperature ...View the full answer

Answered By

Pramendra Kumar Singh
To be very honest, i have only a little experience regarding tutoring, that too was "applied" on my brother.
But regarding education, I am a confident man, just like John Wick. I obtained the highest possible CGPA in class 10th i.e 10CGPA. I was good at all the subjects, but my love for English, Social Studies and Mathematics were on a whole different level.
So for the above mentioned reason, i went for Mathematics for higher studies. Physics became my new love. And then, I ended up cracking JEE Mains, but couldn't go through Advanced. I gave a thought of taking a drop for a year, but my family situation was a bit shaky and a middle class boy couldnt afford to take the so-called "extreme risks".
Thus I gave a state level examination of Chhattisgarh state, where i live(btw). I obtained a rank of 274 out some lakhs (that was a relief back then). So, eventually i got admission into a good college (some say, topmost in our state).
But i, belonging to a class of kids who were brought up with 'being humble' as a centre of their principles, i worked hard. I still am. Electrical engineering, tough nut to crack. But Grind is the other name of Life. So i am still one of you students, working hard, having patience and also, trying to earn some money side-by-side to be free from always asking my parents for money. Believe me, after a certain age, u all will also feel the same way. In my case, i am slowly and steadily becoming allergic to asking for economic help, not atleast for my petrol and other daily expenses.
Also, you may find me using puns ans SARCASM a lot. Thats who i am. I joke a lot. I keep things simple. And, i am a man of 'wisdom' too. So i might employ some of my counselling skills with you too.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Describe what you would observe if you heated the solid at the composition 40. atomic percent Si in Figure 9.26 from 300.C to 1300.C.
-
A strip of aluminum metal is placed into a beaker containing 1.0 M CuSO4. (a) Will a spontaneous reaction occur? (b) If a spontaneous reaction does occur, write the half-reactions and describe what...
-
Describe what you would look for in a reaction involving gases in order to predict the sign of So. Explain.
-
On average, do people prefer a... 6 game package with 300-level, mid-court seats and a $20 gift certificate for $35/seat or a 3 game package with 300-level, corner seats that come with a hotdog and...
-
Jan finished culinary school and wants to start her own pastry business. She has few options, and believes that a sensitivity analysis would help her to make the best business decision. She is...
-
The Gallup poll in Example 6 reported that during March 2011, 60% of Americans favored offshore drilling as a means of reducing U.S. dependence on foreign oil. The poll was based on the responses of...
-
A four cylinder, two-stroke cycle petrol engine develops \(30 \mathrm{~kW}\) at \(2500 \mathrm{rpm}\). The indicated mean effective pressure of each is \(800 \mathrm{kPa}\) and \(\eta_{\text {mech...
-
RL Photography reported net income of $100,000 for 2014. Included in the income statement were depreciation expense of $6,300, patent amortization expense of $4,000, and a gain on disposal of plant...
-
why it is important to have a good first impression and why you must stand out to the interviewer ? As a health care professional, what are the skills you can develop to build positive relationships...
-
A research specialist for a large seafood company investigated bacterial growth on oysters and mussels subjected to three different storage temperatures. Nine cold storage units were available. Three...
-
Suppose the man does not run for 6 months over the winter due to snow on the ground. He resumes running once a week in the spring and records a running time = 12.97 minutes in his first week of...
-
The heat of fusion of water is 6.008 10 3 J mol 1 at its normal melting point of 273.15 K. Calculate the freezing point depression constant K f .
-
Find the intensity of two identical light sources located 0.454 m and 0.538 m, respectively, from a point where the illumination is 8.46m/m2
-
Determine whether the events E and F are independent or dependent. Justify your answer. (a) E: A person living at least 70 years. F: The same person regularly handling venomous snakes. (b) E: A...
-
Design a bandstop elliptic filter satisfying the following specifications: \[\begin{aligned}A_{\mathrm{p}} & =0.5 \mathrm{~dB} \\A_{\mathrm{r}} & =60 \mathrm{~dB} \\\Omega_{\mathrm{p}_{1}} & =40...
-
Suppose you just purchased a digital music player and have put 6 tracks on it. After listening to them you decide that you like 2 of the songs. With the random feature on your player, each of the 6...
-
Until recently, investors purchased bonds almost entirely for income. Over the past 20 years investors have witnessed increased bond price volatility and the advent of junk bonds. Assume that you...
-
Design a lowpass filter satisfying the following specifications: \[\begin{aligned}M(\Omega T)=1.0, & \text { for } 0.0 \Omega_{\mathrm{s}}
-
What is it about each of these decisions that caused them to be listed as the most important business decisions? Propose a recent business decision that is a candidate for this list.
-
On January 1, 2017, McIlroy, Inc., acquired a 60 percent interest in the common stock of Stinson, Inc., for $340,200. Stinson's book value on that date consisted of common stock of $100,000 and...
-
The vapour pressure of benzene between 10C and 30C fits the expression log(p/Torr) = 7.960 1780/(T/K). Calculate (a) The enthalpy of vaporization and (b) The normal boiling point of benzene.
-
The vapour pressure of a liquid in the temperature range 200 K to 260 K was found to fit the expression ln(p/Torr) = 16.255 2501.8/(T/K). Calculate the enthalpy of vaporization of the liquid.
-
Prior to the discovery that freon-12 (CF 2 Cl 2 ) was harmful to the Earths ozone layer, it was frequently used as the dispersing agent in spray cans for hair spray, etc. Its enthalpy of vaporization...
-
a) Give one reason why local Pakistan bus businesses could benefit from Pakistan's crisis. b) Give one reason why local bus businesses could be hurt by the Rupee crisis.
-
A P/E multiple could be high relative to the S&P 500 Index because its high growth rate justifies it. You notice a stock that has an assumed growth rate of 5.43% with a dividend payout ratio of 64.9%...
-
What would be the volume ( cm 3 ) of a piece of Styrofoam if it had a mass of 3 5 0 g , no matter its shape? Show calculation.

Study smarter with the SolutionInn App


