Evaluate the electronic partition function for atomic Fe at 298 K given the following energy levels. Level
Question:
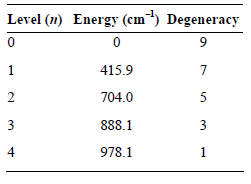
Transcribed Image Text:
Level (1) Energy (cm) Degeneracy 415.9 704.0 888.1 3 978.1 2. 3. 4.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
q ge 9e 7eB4159 cm 0 1 SeB7040cm 4159 cm ...View the full answer

Answered By

Madhur Jain
I have 6 years of rich teaching experience in subjects like Mathematics, Accounting, and Entrance Exams preparation. With my experience, I am able to quickly adapt to the student's level of understanding and make the best use of his time.
I focus on teaching concepts along with the applications and what separates me is the connection I create with my students. I am well qualified for working on complex problems and reaching out to the solutions in minimal time. I was also awarded 'The Best Tutor Award' for 2 consecutive years in my previous job.
Hoping to get to work on some really interesting problems here.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
a. Evaluate the electronic partition function for atomic Si at 298 K given the following energy levels: b. At what temperature will the n = 3 energy level contribute 0.100 to the electronic partition...
-
NO is a well-known example of a molecular system in which excited electronic energy levels are readily accessible at room temperature. Both the ground and excited electronic states are doubly...
-
Evaluate the translational partition function for H 2 confined to a volume of 100 cm 3 at 298 K. Perform the same calculation for N 2 under identical conditions. (Do you need to reevaluate the full...
-
On April 30, the end of the first month of operations, Joplin Company prepared the following income statement, based on the absorption costing concept: Joplin Company Absorption Costing Income...
-
In Exercises 51-54, a. determine the value that the function f approaches as the magnitude of x increases. Is f (x) greater than or less than this value when b. x is positive and large in magnitude...
-
A stratified layer of oil on water is placed between parallel plates. The plates are separated by a distance of \(1 \mathrm{~cm}\) and the oil layer is \(0.4 \mathrm{~cm}\) thick. The water has a...
-
What is a retention and why is it used in managing project work?
-
The Wall Street Journal reported that Juniper Networks, Inc.a maker of company network equipmentplans to offer its more than 1,000 employees the opportunity to reprice their stock options. Junipers...
-
4. A lumber has a nominal dimension of 2x4 inches and is tested for 3-point bending with a span of 4 ft. The wood does not get ruptured until the point load reaches 88 lbs. (Note: The 2-inch side of...
-
For a normal random variable with mean 19,500 and standard deviation 400, find a point of the distribution such that the probability that the random variable will exceed this value is 0.02.
-
Imagine performing the coin-flip experiment of Problem P29.17, but instead of using a fair coin, a weighted coin is employed for which the probability of landing heads is two-fold greater than...
-
Rhodopsin is a biological pigment that serves as the primary photoreceptor in vision (Science 266 [1994]: 422). The chromophore in rhodopsin in retinal, and the absorption spectrum of this species is...
-
List the fossil fuels in the order in which they contribute to world CO 2 emissions.
-
Draw a labor-leisure graph to show the budget constraint (intercepts, slope) for a person that earns $10 wage per hour worked and has 300 hours per month available to allocate between leisure and...
-
Shady Lady sells window coverings to both commercial and residential customers. The following information relates to its budgeted operations for the current year: Revenues Commercial $300,000...
-
How do meritocratic ideals and the realities of systemic inequality coexist in modern societies ? Explain
-
Agassi Company uses a job order cost system in each of its three manufacturing departments. Manufacturing overhead is applied to jobs on the basis of direct labor cost in Department D, direct labor...
-
How do conflict theory and structural functionalism differently interpret the role of institutions in perpetuating or challenging societal inequalities ?
-
Triple J Movers Ltd. is owned by Jacques Ttreault. The company used to be profitable but several new small companies have started to compete with Triple J, offering very low prices that Triple J...
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
Consider the structure of the following compound: (a) When this compound is treated with bromine under conditions that favor monobromination, two stereoisomeric products are obtained. Draw them, and...
-
Name one element that you would expect to exhibit bonding properties similar to boron. Explain?
-
Nicotine is an addictive substance found in tobacco. Identify the hybridization state and geometry of each of the nitrogen atoms in nicotine: C-H N. C- H. Nicotine z: I I-O
-
What role does ethical leadership play in crisis management, and how can leaders ensure that ethical principles guide their actions and communications during times of significant stress, uncertainty,...
-
a) Parameterize the curve of intersection of the surfaces x+y=9 and 2x-y+2=5. Use Maple to plot the surfaces together with the curve of intersection. b) Use Maple to plot the surface f(x,y) = xy c)...
-
Any business that operates across state lines must determine the amount of their income that is subject to tax in each state. Given that there is no uniform approach to apportionment, states use...

Study smarter with the SolutionInn App


