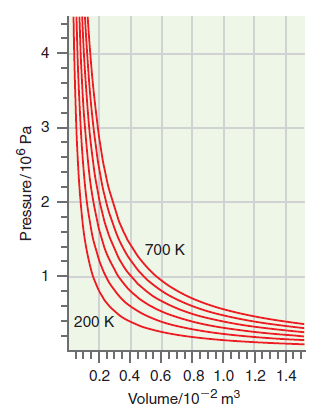
Explain how the ideal gas law can be deduced for the measurements shown in Figures 1.5 and
Question:
Figure 1.5
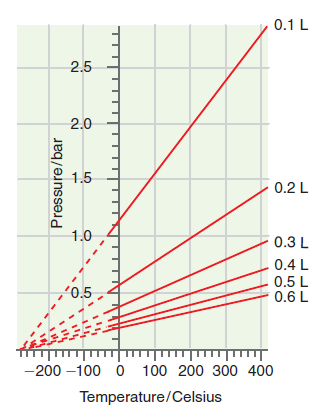
Figure 1.8
Transcribed Image Text:
0.1 L 2.5 2.0 1.5 0.2 L 1.0 0.3 L 0.4 L 0.5 L 0.6 L '0.5 тп -200 –100 0 100 200 300 400 Temperature/Celsius Pressure/bar 700 K 200 K Tпт 0.2 0.4 0.6 0.8 1.0 1.2 1.4 Volume/10-2 m3 3. 2. Pressure/106 Pa
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
Figure 15 shows that at constant volume P increases ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Use the ideal gas law to show that, for an ideal gas at constant pressure, the coefficient of volume expansion is equal to = 1/T, where T is the temperature in kelvins. Compare to Table 13-1 for...
-
Pressure of an ideal gas. It is desired to get the pressure exerted by an ideal gas on a wall by accounting for the rate of momentum transfer from the molecules to the wall.? (a) When a molecule...
-
Adiabatic frictionless processes in an ideal gas (a) A gas that obeys the ideal gas law may deviate appreciably from C p = constant. Hence rework Example 11.4-6 using a molar heat capacity expression...
-
CPA firm brings in a yoga instructor during the tax busy season to help relieve stress of the employees. Which is true about the CPA firm's ability to take a deduction for the yoga instructor's...
-
1) What is HiTech's pool rate for the material-handling activity closest to? 2) What is HiTech's pool rate for the automated machinery activity closest to? 3) What is HiTech's pool rate for the...
-
You are designing an engine that contains several pistons that each move \(150 \mathrm{~mm}\) per stroke. You believe the engine may consume less energy than a traditional engine if many of the...
-
A machine, modeled as a single-degree-of-freedom system, has the following parameters: \(m=900 \mathrm{~kg}, k=7 \mathrm{MN} / \mathrm{m}\), and \(c=135 \mathrm{~N}-\mathrm{s} / \mathrm{m}\). It is...
-
Jennifer Baskiter is president and CEO of Plants&More.com, an Internet company that sells plants and flowers. The success of her startup Internet company has motivated her to expand and create two...
-
The Keyshawn Harris officer earns $10,100 per month. As of May 31, her gross pay was $50,500. The Federal Insurance Contributions Act (FICA) tax rate for Social Security is 6.2% of the first $137,700...
-
For each of the three generic strategies (i.e., cost leadership, differentiation, and focus), which of the four competitive uses of information is most powerful?
-
Compound A has molecular formula C 5 H 10 . Hydroboration- oxidation of compound A produces 2-methylbutan-1-ol. Draw the structure of compound A: Compound A (C,H10) 1) BH, THF 2) H202, NaOH
-
A bowling ball (a) Rolls across a table (b) Falls on the floor. Is the work associated with each part of this process positive, negative, or zero?
-
In Exercises find the derivative of the function. y = 8 arcsin X 4 x 16x 2
-
The case studies titled "Financial Challenges & Recovery," most likely describe what kind of individual: Someone who is just starting out. Someone who has made major financial mistakes. Someone who...
-
What report shows the quantity, asset value, and average cost?
-
Who would be the best market segment for the hat company to pursue if wanting to increase sales revenue?
-
Previous years cash distributions to a shareholder have reduced his/her stock basis to zero in an S corporation. How will any further cash distributions be treated for tax purposes if the company...
-
What updates are made to Trust Accounting for a payment for repairs of a rental property from the rental funds available to the Landlord: What updates are made to Trust Accounting for a payment for...
-
Write the terms of for each function. Evaluate the sum. (x) = x 2 - 1 Ef(x;)Ax, with x1 = 0, x2 = 2, x3 = 4, x4 = 6, and Ax = 0.5, i=1
-
What are the principal alloying elements in SAE 4340 steel?
-
At 18C the total volume V of a solution formed from MgS04 and 1.000 kg of water fits the expression v = 1001.21 + 34.69(x - 0.070)2, where v = V/cm3 and x = blb-1. Calculate the partial molar volumes...
-
The following table gives the mole fraction of methylbenzene (A) in liquid and gaseous mixtures with butanone at equilibrium at 303.15 K and the total pressure p. Take the vapour to be perfect and...
-
Use the Gibbs-Duhem equation to show that the partial molar volume (or any partial molar property) of a component B can be obtained if the partial molar volume (or other property) of A is known for...
-
Multiplication of signed operands, which generate a double-length product in the 2's-complement 100 11 MULTIPLICAND (-13) 0 10 1 1 MULTIPLIER (+11) number system. The general strategy is the...
-
panel = Drawing Panel (160, 160) for i in range (0, 10): panel.draw rectangle (20, 20 + 100 10 * 1, 10 i, 10) Write variations of the above program that draw the figures at the lower right as output....
-
John estimated the following cash flows (in $) for a project: A B 1 Year Cash flow 2 0 -5,700 3 1 1,325 4 2 2,148 5 3 3,528 The required return for the project is 8%. What is the IRR for the project?

Study smarter with the SolutionInn App


