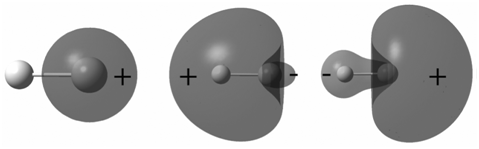
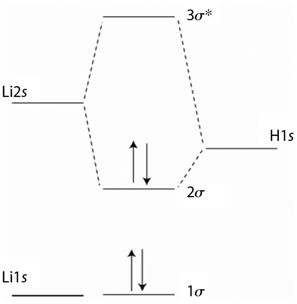
Explain the difference in the appearance of the MOs in Problem P23.13 with those for HF. Based
Question:
Transcribed Image Text:
За* Li2s H1s 20 Lils lo
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The MOs on LiH are more delocalized than on HF so that ...View the full answer

Answered By

Patrick Busaka
I am a result oriented and motivated person with passion for challenges because they provide me an opportunity to grow professionally.
5.00+
38+ Reviews
58+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain the difference in the percentages of the products in these two hydroborationreactions: CH3 CH, QH CH, 1) BH3. THF 2) H,O,, NAOH CH,CH-CHCH-CH; + CH;CHCH,CHCH3 (43%) CH,CHCH=CHCH, (57%) 1)...
-
The distance between Li+ and Cl- is 257 pm in solid LiCl and 203 pm in a LiCl unit in the gas phase. Explain the difference in the bond lengths?
-
Explain the difference in the melting points of the following compounds: Only one of the two can form intramolecular hydrogen bonds? NO2 NO OH OH m.p. 115C m.p. 45C
-
On September 30, 2021, Antagonia Real Estate Limited's general ledger showed the following balances: Accounts payable Accounts receivable Advertising expenses Cash Common shares Income tax expense...
-
In Exercises 1-4, match the function with its graph. State the period of the function. [The graphs are labeled (a), (b), (c), (d), (e), and (f).] a. b. c. d. e. f. 1. y = sec 2x 2. y = tan x / 2 3. y...
-
Below is a list of transactions completed by Direct Fashion during 2020. Ignore GST. For each transaction, indicate (a) the section (i.e. operating, investing or financing) of the statement of cash...
-
How to interview clients?
-
In the 1990s, Pfizer, Inc., developed a new antibiotic called Trovan (trovafloxacin mesylate). Tests showed that in animals Trovan had life-threatening side effects, including joint disease, abnormal...
-
X-Sal-Ent Corp produces frozen dinners. The company has a policy that ending inventory of frozen dinners must be 30% of the following month's expected sales. Required: January February March Unit...
-
Accounting Case 20-10 Auditing Logistical Logistics Background Logistical Logistics Inc. (Logistical Logistics or the Company) provid O The contract identifies the shipping containers and acceptable...
-
Sketch out a molecular orbital energy diagram for CO and place the electrons in the levels appropriate for the ground state. The AO ionization energies are O2s: 32.3 eV; O2p: 15.8 eV; C2s: 19.4 eV;...
-
Calculate the bond order in each of the following species. Predict which of the two species in the following pairs has the higher vibrational frequency: a. Li 2 or Li + 2 b. C 2 or C + 2 c. O 2 or O...
-
Whole foods to become first national supermarket to ban plastic straws Whole Foods is set to completely ban plastic straws and will introduce paper straws across all its stores in the U.S., Canada...
-
Discuss the impact on the present value (PV) of a future lump sum as either the discount rate or the number of compounding periods per year increases .
-
You buy a six-year, 8 percent savings certificate for $10,000. If the interest is compounded semiannually , what will be its value at maturity? Explain the effect that causes the ending balance to...
-
ARU systems provide several options for callers that identify specific departments or services that callers can be connected with directly. What kinds of caller options might be appropriate for an...
-
Year-to-date, Conglomco has earned a 1.64 percent return, Supercorp has earned a 5.69 percent return, and Megaorg has earned a 0.23 percent return. If your portfolio is made up of 40 percent...
-
6. Consider a binomial option pricing setting in which u=1.15, d=0.94, rf=1.06 and So=$15.00. What is the Binomial Option Pricing Model value of a call with a strike price of $16?
-
Today is September 16, Year 2. You, CPA, work for Garcia & Garcia LLP, a medium-sized firm located in Montreal. Jules Garcia calls you into his office. "CPA, I have a very special engagement for you....
-
You purchase a bond with a coupon rate of 6.7 percent, a par value $1,000, and a clean price of $905. Assume a par value of $1,000. If the next semiannual coupon payment is due in two months, what is...
-
Is arsenic-doped germanium a p-type or n-type semiconductor?
-
Calculate the packing fractions of (a) A primitive cubic lattice, (b) A bcc unit cell, (c) An fcc unit cell.
-
Poissons ratio for polyethylene is 0.45. What change in volume takes place when a cube of polyethylene of volume 1.0 cm 3 is subjected to a uniaxial stress that produces a strain of 1.0 per cent?
-
A skier glides down a frictionless hill of 100 meters high, then ascends another hill, with a height of 90 meters, as shown in the figure below. a) What is the speed of the skier when they reach the...
-
The atoms in a nickel crystal vibrate as harmonic oscillators with an angular frequency of 5.5x 1013 rad/s. The mass of a nickel atom is 9.75 x 10-26 kg. What is the difference in energy between...
-
Ballistic pendula are used by forensic analysts to calculate the velocity of a bullet. The bullet is shot into a target suspended by a long, thin, narrow rod; the degree to which the pendulum is...

Study smarter with the SolutionInn App


