Explain why the fluorescence and absorption groups of peaks in Figure 25.10 are shifted and show mirror
Question:
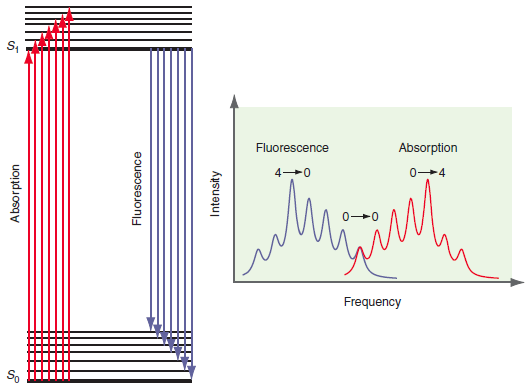
Transcribed Image Text:
Fluorescence Absorption 0-4 Frequency So Absorption Fluorescence Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
The absorption event is initiated from the n 0 vibrational level of the ground state The rela...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The fluorescence spectrum of anthracene vapour shows a series of peaks of increasing intensity with individual maxima at 440 nm, 410 nm, 390 nm, and 370 nm followed by a sharp cut-off at shorter...
-
1. G. Johnson and colleagues have analyzed the MAP kinase cascade in which MEKK2 participates in mammalian cells. By a yeast two-hybrid screen (see Chapter 7), MEKK2 was found to bind MEK5, which can...
-
What is the difference between a fluorescence excitation spectrum and a fluorescence emission spectrum? Which one resembles an absorption spectrum?
-
The conjugate base of diethyl malonate can serve as a nucleophile to attack a wide range of electrophiles. Identify the product that is formed when the conjugate base of diethyl malonate reacts with...
-
A guy wire runs from the ground to a cell tower. The wire is attached to the cell tower 150 feet above the ground. The angle formed between the wire and the ground is 43° (see figure). (a) How...
-
What is the pro forma statement, and how important is it for a business?
-
What is a system development process model?
-
Nick Waege started his own consulting firm, Waegelein Consulting, on June 1, 2010. The trial balance at June 30 is as follows. In addition to those accounts listed on the trial balance, the chart of...
-
During 2024, a company sells 365 units of inventory for $95 each. The company has the following inventory purchase transactions for 2024: Number of Date January 1 May 5 Transaction Beginning...
-
Mohan is a sole trader who does not maintain a full set of accounting records. He was able to provide the following information: Fixtures should be depreciated by 10% per annum on the cost of...
-
What aspect of the confocal microscope makes single-molecule spectroscopy in solutions possible?
-
The rate of fluorescence is higher than that for phosphorescence. Can you explain this fact?
-
The Dow Jones Industrial Average on July 20, 2016 was 18,580 and the price of a September 185 (European) call option on the index was $3.35. Use the DerivaGem software to calculate the implied...
-
How does Benjamin's critique of mass culture different from both conservative and critical theoretic responses?
-
Pharoah Corporation earned revenues of $224800, incurred expenses of $233000, and paid dividends of $4350 during the current year. When Income Summary is closed to Retained Earnings What is the debit...
-
The overall purpose of research is to add to the body of knowledge using an ethical and systematic process. For each article: Q.1 For each article, discuss how the researchers would have ensured...
-
Harris Fabrics computes its plantwide predetermined overhead rate annually on the basis of direct labor - hours. At the beginning of the year, it estimated that 3 1 , 0 0 0 direct labor - hours would...
-
What is the difference between repressive state apparatuses and ideological state apparatuses?
-
Cedar Corporation reported a net operating loss in 2018 of $25,000,000. In 2019, Cedar reported taxable income before any NOL carryovers of $20,000,000. What is Cedars taxable income in 2019 and any...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Calculate the maximum (zero-current) potential difference of a nickelcadmium cell, and the maximum possible power output when 100 mA is drawn at 25C.
-
Derive an expression for the current density at an electrode where the rate process is diffusion-controlled and c is known. Sketch the form of j/j L as a function of c . What changes occur if anion...
-
Can magnesium be deposited on a zinc electrode from a unit activity acid solution at 25C?
-
Design a power transmission system to connect a DC motor to its load. The motor is given in the motor catalogue as a BM1400 motor. You must create a transmission which will connect to this motor and...
-
SDJ, Incorporated, has net working capital of $3,320, current liabilities of $4,550, and inventory of $4,600. a. What is the current ratio? b. What is the quick ratio?
-
Calculate the final selling price to the nearest cent and markdown percent to the nearest hundredth percent: Original selling price $4,900 First markdown 20% Second markdown 10% Markup 12% Final...

Study smarter with the SolutionInn App


