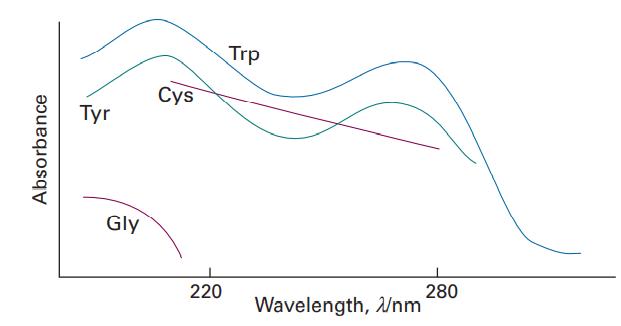
Figure 13.2 shows the UV-visible absorption spectra of a selection of amino acids. Suggest reasons for their
Question:
Figure 13.2 shows the UV-visible absorption spectra of a selection of amino acids. Suggest reasons for their different appearances in terms of the structures of the molecules.
Data in Figure 13.2
Transcribed Image Text:
Absorbance Tyr Gly Cys 220 Trp Wavelength, Wnm 280
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
Answer The different appearances of the UVvisible absorption spectra ...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The 20 naturally occurring amino acids (See the following table) are all l amino acids, and they all have the S configuration, with the exception of glycine (which lacks a chirality center) and...
-
The UV spectrum of 1-phenylprop-2-en-1-ol shows an intense absorption at 220 nm and a weaker absorption at 258 nm. When this compound is treated with dilute sulfuric acid, it rearranges to an isomer...
-
A mixture of amino acids that do not separate sufficiently when a single technique is used can often be separated by two-dimensional chromatography. In this technique, the mixture of amino acids is...
-
aj b) If the magnitude of the force F is 700 N, what is the value of the internal moment at point A in N.m? d) 0.5 m 540 180 210 A 0.8 m 240 Bo birak F A
-
What do the terms debit and credit mean?
-
A guy wire is helping to support a utility pole in Ilsis backyard, as shown in the diagram. The point U is located 3 feet below the top of the pole. a. What is the angle of elevation of the guy wire?...
-
Investigate the file search facility on your computer. Which type of search method do you think it uses? Why do you think this partic ular search method was chosen? What problems could this approach...
-
(Three Differences, Multiple Rates, Future Taxable Income) During 2010, Graham Co.'s first year of operations, the company reports pretax financial income of $250,000. Graham's enacted tax rate is...
-
b. A bank sells a three against six $4,000,000 FRA for athree-month period beginning three months from today and ending sixmonths from today. The purpose of the FRA is to cover the interestrate risk...
-
You are the owner of a small business that manages the program, food, and merchandise sales at the Excel Center, where the local professional basketball team plays. In addition to basketball games,...
-
3-Buten-2-one (3) has a strong absorption at 213nm and a weaker absorption at 320nm. Justify these features and assign the ultraviolet absorption transitions. 3 3-Buten-2-one
-
Estimate the magnitude of the transition dipole moment of a chargetransfer transition modelled as the migration of an electron from a H1s orbital on one atom to another H1s orbital on an atom a...
-
Using a required rate of return equal to 12 percent, compute the modified internal rate of return (MIRR) for a project that cots $82,000 and is expected to generate $35,000, $70,000, and -$10,450,...
-
Show that: (a) If \(H(z)\) is a Type I filter, then \(H(-z)\) is Type I. (b) If \(H(z)\) is a Type II filter, then \(H(-z)\) is Type IV. (c) If \(H(z)\) is a Type III filter, then \(H(-z)\) is Type...
-
Calculate DVA in Example 24.6. Assume that default can happen in the middle of each month. The default probability of the bank is 0.001 per month for the two years and the recovery rate in the event...
-
Calculate the price of a six-month European put option on the spot value of an index. The six-month forward price of the index is 1,400, the strike price is 1,450, the risk-free rate is 5%, and the...
-
Xanadu Mining is considering three mutually exclusive alternatives, as shown in the table below. MARR is 10 percent/year. a. What is the present worth of each alternative? b. Which alternative should...
-
What is the formula for the delta of a call option on a stock that provides no income?
-
A particular form of electromagnetic radiation has a frequency of 8.11 1014 Hz. (a) What is its wavelength in nanometers? In meters? (b) To what region of the electromagnetic spectrum would you...
-
In Exercises, find the equation of the tangent line at the given point on each curve. 2y 2 - x = 4; (16, 2)
-
Explain without using equations why (H /P)T 0H 0P is generally small for a real gas.
-
Why is it reasonable to write dH C P dT + VdP for a liquid or solid sample?
-
Refer to Figure 1.10 and explain why (U/V) T is generally small for a real gas. Figure 1.10 Ideal gas Real gas Tranlaition rv-0 (4)A
-
Suppose Stocks A, B and C are the only three component stocks in a benchmark index. The number of shares outstanding of Stocks A, B and C are 369,000 shares, 327,000 shares, and 240,000 shares,...
-
Botanists have determined that some species of weed grow in a circular pattern. For one such species, the area A , in square meters, can be approximated by A ( t ) = 0.004 t 2 , where t is the time...
-
Book Print ciences Gallatin Carpet Cleaning is a small, family-owned business operating out of Bozeman, Montana. For its services, the company has always charged a flat fee per hundred square feet of...

Study smarter with the SolutionInn App


