Figure 16.17 shows that atomic level resolution is only attainable in the repulsive portion of the tipsurface
Question:
Figure 16.17
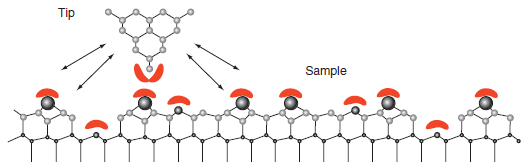
Transcribed Image Text:
Tip Sample
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The observation shows that the repulsive part ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Why is atomic level resolution obtained on pentacene in the AFM mode as shown in Figure 16.18, but not in the STM mode? Figure 16.18 1.3 A OA +1Hz -2Hz D 20A 5A -7Hz. -5Hz.
-
The DNA double helix (Figure 24.30) at the atomic level looks like a twisted ladder, where the "rungs" of the ladder consist of molecules that are hydrogen-bonded together. Sugar and phosphate groups...
-
The spent fuel elements from a fission reactor are much more intensely radioactive than the original fuel elements. (a) What does this tell you about the products of the fission process in...
-
What is your debt payments-to-income ratio if your debt payments total $684 and your net income is $2,000 per month?
-
Use the graph to find the projection of u onto v. (The terminal points of the vectors in standard position are given.) Use the formula for the projection of u onto v to verify your result. 1. 2. 3....
-
(a) Would you expect the probability that a randomly selected car will need major repairs in the next year to be smaller, remain the same, or increase if you are told it already has high mileage?...
-
a. What information is required to determine sample size in a basic variables sampling plan of inventory balances? b. Assuming the auditor has calculated the estimated population value, what...
-
Jones Co. is in a technology-intensive industry. Recently, one of its competitors introduced a new product with technology that might render obsolete some of Joness inventory. The accounting staff...
-
Today, you observe the following spot rates: Maturity (years) Spot rate 0.5 4.0% 1 4.3% 1.5 5.1% 2 5.5% (All rates are annualized on a semi-annual bond equivalent yield basis and so should be your...
-
Match each equation to one of the graphs. a. 9x2 + 4y2 - 36 = 0 b. x2 - 4y2 - 8x = 0 c. 3x2 - 30x + 5y + 55 = 0 d. x2 + y2 + 2x - 6y - 15 = 0 10 10 10
-
The reflection probability from a step potential was calculated for E > V 0 in Section 16.5. Is Equation (16.18) valid for E < V 0 ? What information can you extract from Figure 16.1 that will allow...
-
Why were quantum dots emitting in the near infrared region used for the surgery experiment shown in Figure 16.25? Figure 16.25 Color video 5 min post-injection NIR fluorescence 5 min post-injection...
-
1. Andersons bank requires a compensating balance of $3 million. How much additional funds can be freed up for investment in fixed assets if the firm reduces its cash balance to the minimum required...
-
The real-life case is the case of Nazanin Zaghari-Ratcliffe. Focus on an international perspective. Examples of the theories and concepts include Discriminatory justice and Intersectionality. These...
-
Special Education Law Module 6 Assignment: State and Federal Law and IEP requirements Geneva is a thirteen-year-old student with behavior issues and a learning disability. She has had an IEP for the...
-
The following is a portion of the condensed income statement for Rowan, Incorporated, a manufacturer of plastic containers: Net sales Less: Cost of goods sold: Inventory, January 1 Net purchases...
-
Pharoah Company has 496000 shares of $10 par value common stock outstanding. During the year Pharoah declared a 15% stock dividend when the market price of the stock was $34 per share. Three months...
-
Look for a corpotate strategy such as vertical integration and diversification, differentiation, innovation, etc. that about Beauty company Glossier Inc., and describe the strategy and why it is...
-
Use the accompanying graph of y = f(x). Find
-
6. (Potential Energy and Conservation of Energy) What should be the spring constant k of a spring designed to bring a 1200-kg car to rest from a speed of 95 km/h so that the occupants undergo a...
-
Molecular orbital calculations based on semi-empirical, ab initio, and DFT methods describe the spectroscopic properties of conjugated molecules better than simple Hckel theory. (a) Use the...
-
Use an appropriate semi-empirical method to compute the equilibrium bond lengths and standard enthalpies of formation of (a) ethanol, C 2 H 5 OH, (b) 1,4-dichlorobenzene, C 6 H 4 Cl 2 . Compare to...
-
Use appropriate electronic structure software and basis sets of your or your instructors choosing, perform self-consistent field calculations for the ground electronic states of H 2 and F 2 ....
-
Question 3 (20 marks) Hollyford Corporation expects to have earnings this coming year of $3 per share . Hollyford plans to retain all of its earnings for the next two years . For the subsequent two...
-
On April 1, 2022 Sport Mart Inc.'s board of directors declared a 10% share dividend when the market price per share was $10. On November 15, 2022, the board of directors declared a 4:1 share split....
-
To record a receipt of payment in January for services not performed: $250 for February and $450 for March, the following is included in the journal entry Debit cash for $700, credit revenue for $700...

Study smarter with the SolutionInn App


