Figure 5.2 shows the phase diagram for two partially miscible liquids, which can be taken to be
Question:
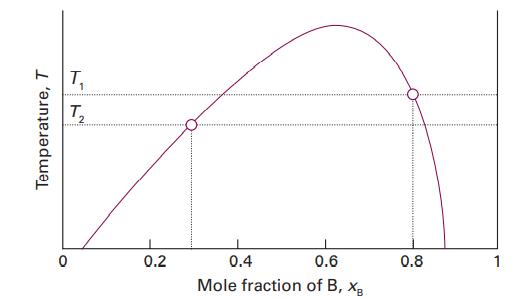
Figure 5.2 shows the phase diagram for two partially miscible liquids, which can be taken to be that for water (A) and 2-methyl-1-propanol (B). Describe what will be observed when a mixture of composition xB =0.8 is heated, at each stage giving the number, composition, and relative amounts of the phases present.
Data in Figure 5.2
Transcribed Image Text:
Temperature, T T₁ 1 T₂ 0.2 0.4 0.6 Mole fraction of B, XB 0.8 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
As the mixture is heated the following will be observed 1 At t...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Figure 5.8 shows the phase diagram for the ternary system NH 4 Cl/ (NH 4 ) 2 SO 4 /H 2 O at 25 C. Identify the number of phases present for mixtures of compositions (i) (0.2, 0.4, 0.4), (ii) (0.4,...
-
What steps can be taken to make controlling costs easier? Can these steps also be used to control other project parameters, such as scope?
-
What actions can be taken to reduce derived demand?
-
Khairul has been working with Tun Hamid Energy Bhd since 1 February 2009 as a Human Resource Executive. Beginning 1 January 2014, he was promoted as Human Resource Manager. Details of his...
-
The rules related to the capitalization of interest costs into the original carrying value of PPE are generally similar under U. S. GAP and IFRS. While there are some differences, most notably in how...
-
You are provided with the following information for Aylesworth Inc. for the month ended October 31, 2012. Aylesworth uses a periodic method for inventory. Instructions(a) Calculate (i) ending...
-
The following MINITAB output presents a 95% confidence interval for the mean ozone level on days when the relative humidity is 60%, and a 95% prediction interval for the ozone level on a particular...
-
The TCM Petroleum Corporation is an integrated oil company headquartered in Fort Worth, Texas. Historical income statements for 2014 and 2015 are found below (dollar figures are in the millions): In...
-
The Baltimore Arena, formerly known as Royal Farms Arena, is the city's largest indoor sports and performance venue and is undergoing a $150M renovation, including overhauled concourses, 30 to 40 new...
-
Asian Transit Marine Corporation (Lessee) leases a forklift from NDP Leasing Corp. (Lessor). The lease, signed on January 1, 2019, specifies that NDP grants right-of-use of the lift to Asian Transit....
-
Suggest an interpretation of the additional terms in extended versions of the DebyeHckel limiting law.
-
Show that the osmotic pressure of a real solution is given by V=RT ln aA. Go on to show that, provided the concentration of the solution is low, this expression takes the form V=RT[B] and hence that...
-
1. Is SAP's corporate structure hindering its competitive advantage? 2. What corporate strategies has SAP used to remain competitive? 3. How has SAP's business-level strategies sustained them...
-
The partnership of Ace, Jack, and Spade has been in business for 25 years. On December 31, 20X5, Spade decided to retire. The partnership balance sheet reported the following capital balances for...
-
1. Given the refrigeration system, draw the complete actual Ph diagram and compute for total work of compression in KW. EV 11 37C CONDENSER io kw 10C 12 EVAPORATOR 13 FLASH PRV 14 TANK AND...
-
What is h'(x) when h(x) = log7 8z+4 2672
-
The following restaurants: Buca di Beppo - Italian, Denny's Restaurant - American, Panda Restaurant - Chinese. Can you pls. explain the symbolism , scheme , and kind of messaging does the font style...
-
Q.1. Let's practice the concept of polyphase decomposition of a sequence. Consider the following sequence: [10] CLO 3 h[n] = 2n + 1 0n7 a) Consider M = 4 and pass h[n] through the block diagram shown...
-
(a) Let K and L be symmetric n x n matrices. Prove that xTKx = xTx for all x Rn if and only if K = L. (b) Find an example of two non-sy mmetric matrices K L such that xTKx = xTLx for all X Rn.
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
One mole of H 2 O(l) is super cooled to 3.75C at 1 bar pressure. The freezing temperature of water at this pressure is 0.00C. The transformation H 2 O(l) H 2 O(s) is suddenly observed to occur. By...
-
An athlete at high performance inhales ~3.75 L of air at 1.0 atm and 298 K at a respiration rate of 32 breaths per minute. If the exhaled and inhaled air contain 15.3 and 20.9% by volume of oxygen,...
-
The temperature of 1.75 moles of an ideal gas increases from 10.2C to 48.6C as the gas is compressed adiabatically. Calculate q, w, U, and H for this process assuming that C V ,m = 3/2 R.
-
Jeremy Pruitt Ltd is considering the replacement of a delivery truck. The current truck could last for three more years. Operating costs are 5000 per year. We are currently depreciating it at 4000...
-
As a long-term investment at the beginning of the 2024 fiscal year, Florists International purchased 30% of Nursery Supplies Incorporated's 8 million shares of capital stock for $30 million. The fair...
-
PJ Ltd Co. has been contracted to supply two products S & T worth 500 pieces, the joint cost function for the two varieties is given as: C = 100S2 +150T2. The quantity of S and T are not specified...

Study smarter with the SolutionInn App


