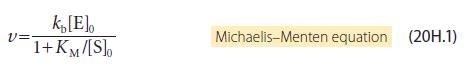
For many enzymes, the mechanism of action involves the formation of two intermediates: Show that the rate
Question:
For many enzymes, the mechanism of action involves the formation of two intermediates:

Show that the rate of formation of product has the same form as that shown in eqn 20H.1, but with vmax and KM given by
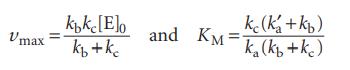
Data in eqn 20H.1
Transcribed Image Text:
E+S →ES ES →E+S ES ES' ES' → E+P v=k₂[E][S] v=k₁ [ES] v=k₁[ES] v=k [ES']
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
solution The rate of formation of product in the given enzymatic rea...View the full answer

Answered By

Brian Nambale
As someone who loves to read books and discuss ideas with friends, I have developed a strong foundation of good knowledge that allows me to understand complex topics well, making me confident in my ability to deliver subjects effectively when teaching children.Engaging in thought-provoking discussions with friends helps me deepen my understanding and apply good thinking to expand my knowledge in various subjects, which I am eager to well when teaching children.During my college years, I acquired 5 years of tutoring experience, which not only helped me to support myself financially but also provided me with an opportunity to share my knowledge, inspire students, and make a positive impact on their lives. Through this experience, I developed strong teaching and communication skills, which have served me well throughout my academic and professional journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Describe the mechanism of action for addictive drugs such as cocaine.
-
The mechanism of glycoside formation is the same as the second part of the mechanism for acetal formation. Propose a mechanism for the formation of methyl -D-glucopyranoside.
-
What's the mechanism of action of trizepatide, the new diabetes 2 drug? and how does it differ from GLP-1 agonists?
-
Why should every credit application be signed by the applicant or an authorized officer of the applicant? There are several factors that influence the setting of the terms and conditions of a sale....
-
A written hospital policy prohibited solicitation and distribution in all patient care areas, including hallways adjacent to patients rooms and other areas frequented by patients. The policy...
-
Swifty Maintenance Corp. has a $305 monthly contract with Crane Treat Inc. for general maintenance services. Swifty invoices Crane on the first of the month for services that it performed in the...
-
Identify the key elements of effective project cost management.
-
1. Draw a simple floor plan for New Century Wellness group and include the placement of all network nodes including the placement of a server, and network equipment. How many ports will your switch...
-
Compile a list of international HR mistakes has made so far. How would you have gone about hiring a European sales manager? Why?
-
Compare the statement of revenues, expenditures, and changes in fund balance with the schedule of revenues, expenditures, and changes in fund balance budget and actual, providing both similarities...
-
The mechanism of a composite reaction consists of a fast preequilibrium step with forward and reverse activation energies of 27 kJmol 1 and 35 kJmol 1 , respectively, followed by an elementary step...
-
The rate of a chemical reaction is found to triple when the temperature is raised from 24 C to 49 C. Determine the activation energy.
-
Find a value of x such that 3 - dt = Cx 1 - dt. 1/4 t
-
With regard to thickness and length, what do you think chromosomes would look like if you microscopically examined them during interphase? How would that compare with their appearance during...
-
Discuss the types of experimental observations that Lyon brought together in proposing her hypothesis concerning X-chromosome inactivation. In your own words, explain how these observations were...
-
Three errors have been found in the records of T O Dorrigo and require correction by general journal. a The purchases account was used in error when a new photocopier (office equipment) was purchased...
-
Lets suppose you were looking through a vial of fruit flies in your laboratory and noticed a male fly with pink eyes. What crosses would you make to determine if the pink allele is an X-linked gene?...
-
What is the fastest-growing region for marketing research? What five markets are currently the largest in the world?
-
A compound made up of C, H, and Cl contains 55.0 percent Cl by mass. If 9.00 g of the compound contain 4.19 1023 H atoms, what is the empirical formula of the compound?
-
Government is advised to tax goods whose demand curves are inelastic if the goal is to raise tax revenues. If the goal is to discourage consumption, then it ought to tax goods whose demand curves are...
-
Calculate the expectation value (r r) 2 if the H atom wave function is 100(r).
-
In spherical coordinates, z = r cos. Calculate z and z 2 for the H atom in its ground state. Without doing the calculation, what would you expect for x and y, and x 2 and y 2 ? Why?
-
The force acting between the electron and the proton in the H atom is given by F = e 2 / 4 0 r 2 . Calculate the expectation value F for the 1s and 2p z states of the H atom in terms of e, 0 , and a...
-
Elaborate on "The Value of Learning of Applied Research Methods in Sports Management or Kinesiology" as another section of your Applied Research Methods in Sports Management. source:...
-
1. A entered into a contract with B, who is a minor. Can A later asks for the invalidation of the contract by reason of the minority of B? 10 points 2. A, the seller, and B, the buyer, agreed for the...
-
Prepare journal entries to record transactions a through h. a. Raw materials purchased on credit, $108,000. b. Direct materials used, $44,000. Indirect materials used, $19,800. c. Direct labor used,...

Study smarter with the SolutionInn App


