From the following data, calculate ÎH o R,391.4 K for the reaction CH 3 COOH(g) + 2O
Question:
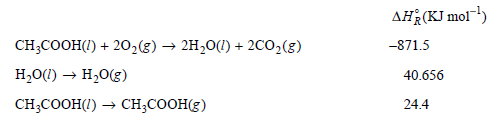
Values for ΔHoR for the first two reactions are at 298.15 K, and for the third reaction at 391.4 K.

Transcribed Image Text:
AH(KJ mol) CH;COOH(I) + 20,(g) → 2H20(1) + 2C0,(g) H,0(1) → H,0(g) CH,CООН() — сH,CОOHE) -871.5 40.656 24.4 Substance CH-COон() 0,(g) co:(g) Н.0() H,0(g) 14.9 3.53 4.46 9.055 4.038 |CP,m/R
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
The desired overall reaction can be related to the individual reactions f...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From the following data, Calculate the enthalpy change for the reaction 2C(graphite) + 3H2(g) - C2H6(g) C(graphite) 02(g)CO2(8) H208)02(8) H0U) 2C2Hs (g)702(g)4CO2(g) 6H2O(l) AHn393.5 kJ/mol...
-
From the following data for the first-order gas-phase isomerization of CH3NC at 215 oC, calculate the first-order rate constant and half-life for the reaction: Time (s) Pressure CH3NC (torr)...
-
Rate constants for a reaction were determined at five temperatures. From the following data, calculate the experimental energy of activation and then calculate G¡, H¡, and S¡ for...
-
In schema normalisation, is Boyce-Codd Normal Form (BCNF) always to be preferred over 3rd Normal Form (4NF)? Explain your answer. [5 marks] 8 (TURN OVER) CST.2004.7.4 8 Economics, Law and Ethics (a)...
-
Thompson Company uses a standard cost system for its single product. The following data are available: Actual experience for the current year: Purchases of raw materials (11,000 yards at $11.00 per...
-
Smile Maids, Inc.s trial balance on February 28, 2013, shows the following: Smile Maids, Inc. completed the following transactions in March: Mar 2 Signed a contract worth $1,200/month for a year to...
-
A pitcher can pitch a "curve ball" by putting sufficient spin on the ball when it is thrown. A ball that has absolutely no spin will follow a "straight" path. A ball that is pitched with a very small...
-
Beaty Company has the following internal control procedures over cash receipts. Identify the internal control principle that is applicable to each procedure. (a) All over-the-counter receipts are...
-
A projectile if fired from 19.6m above the ground with an initial speed of 55.6m/s at an angle of 41.2 degrees above the horizontal. a) Determine the time necessary for the projectile to reach its...
-
1. A Truck is purchased for $200,000 and is planned to have an operating life of 8 years working 5,000 hours per year. What is the hourly owning cost of this machine, assuming the company requires a...
-
Predict the major product obtained upon radical bromination of each of the following compounds: (a) (b) (c)
-
A cylindrical vessel with rigid adiabatic walls is separated into two parts by a frictionless adiabatic piston. Each part contains 45.0 L of an ideal monatomic gas with C V ,m = 3/2R. Initially, T i...
-
Consider the following independent jobs. Overhead is applied in Department 1 at the rate of $6 per direct labor hour. Overhead is applied in Department 2 at the rate of $8 per machine hour. Direct...
-
Look up the names of the largest contractors reported by ENR in its three main categories: Heavy and highway, nonresidential building, and multiunit housing. Notice that the list also includes their...
-
What is a possible endogeneity problem in the Fama and French asset pricing tests of the three-factor model? How can we reduce this problem in asset pricing tests?
-
Normally, joint tests that the intercepts (i s) equal zero are in-sample tests. What is a criticism of this testing approach?
-
If the CAPM is dead, and asset prices are better explained by multiple factors, what theoretical support for multifactor models exists?
-
Explain the concepts of event date, event time, event window, and estimation window.
-
Regard y as the independent variable and x as the dependent variable and use implicit differentiation to find dx/dy. y sec x = x tan y
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
Some of the selection rules for hydrogenic atoms were derived in justification 10.4. Complete the derivation by considering the x- and y components of the electric dipole moment operator.
-
The wave function of a many-electron closed-shell atom can expressed as a Slater determinant (Section 10Ab). A useful property of determinants is that interchanging any two rows or columns changes...
-
The distribution of isotopes of an element may yield clues about the nuclear reactions that occur in the interior of a star. Show that it is possible to use spectroscopy to confirm the presence of...
-
Carmaker produces small cars. As production emits pollution, Carmaker needs to buy pollution permits. It also needs to hire labour. Carmaker's production function is given by: (1) q = min{0.05L,...
-
Describe the three key differences between the North American Free Trade Agreement (NAFTA) NAFTA and United States-Mexico-Canada Agreement (CUSMA). Support your answer by providing specific examples.
-
Discuss what is Brexit. Why did the United Kingdom decided to leave the European Union? What is the impact of trade between the U.K. and the E.U. post-Brexit?

Study smarter with the SolutionInn App


