From the following data, derive the absolute entropy of crystalline glycine at T = 300.K. You can
Question:
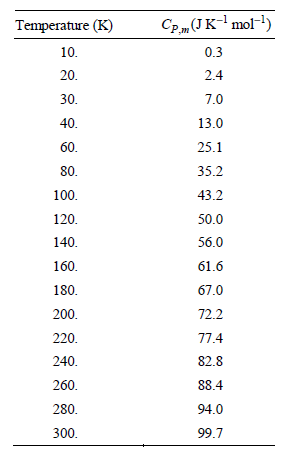
Transcribed Image Text:
CP„(JK-mol-) Temperature (K) 10. 0.3 20. 2.4 30. 7.0 13.0 40. 60. 25.1 35.2 80. 100. 43.2 50.0 120. 140. 56.0 160. 61.6 180. 67.0 72.2 200. 220. 77.4 82.8 240. 260. 88.4 280. 94.0 99.7 300.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
The line in the graph above of C Pm vertical axis against T is the best fit to the ...View the full answer

Answered By

Pranav Makode
I am a bachelor students studying at professor ram meghe institute of technology and research. I have a great experience of being an expert. I have worked as an expert at helloexperts and solvelancer as a part time job. I have also worked as a doubt solver at ICAD SCHOOL OF LEARNING, which is in Amravati city. I have also worked as an Freelancer.
I have great experience of helping students, as described above. I can help any students in a most simple and understandable way. I will not give you have any chance for complaint. You will be greatfull to accept me as an expert.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
From the following data, determine f Ho for diborane, B2H6 (g), at 298 K: (I) B2H6 (g) + 3 O2 (g) B2O3(s) + 3 H20 (g) t Ho=-1941 kJ mol-1 (2) 2 B(s) + t Oh) B2O3(S) t, Ho = -2368 kJ mol-1 (3) H2 (g)...
-
From the following data for liquid nitric acid, determine its heat of vaporization and normal boiling point. Temperature C) Vapor Pressure (mm Hg) 10. 20. 30. 40. 50. 80. 14.4 26.6 47.9 81.3 133 208...
-
From the following data for three prospective fuels, calculate which could provide the most energy per unit volume: Density at 20 C Molar Enthalpy of Combustion Fuel (g/cm (kJ/mol) Nitroethane, C2H...
-
Understand the content theories of motivation.
-
Why are slow-moving items dangerous to the small business? What can be done to liquidate them from inventory?
-
A 48-kg acrobat must jump high and land on his brother's shoulders. To accomplish this, she leaps from a crouched position to a height where her center of mass is \(1.60 \mathrm{~m}\) above the...
-
Diagram the flow of data within general accounting in recording cash receipts transactions.
-
Security Service Companys Accounts Receivable account shows a debit balance of $104,000 at the end of the year. An aging analysis of the individual accounts indicates estimated uncollectible accounts...
-
Refer to the annual report of BHP annual report 2023 (including the table of contents) and answer the following questions: . Review all the sections within the selected company's annual report that...
-
In a subway station, there are exactly enough customers on the platform to fill three trains. The arrival time of the nth train is X1+ + Xn where X1, X2 . . . are iid exponential random variables...
-
How many finished units per day can the current assembly process produce? Where are the bottlenecks?
-
If the process needed to make 18 units per day, what should you do?
-
The December 31, 2024, unadjusted trial balance for Demon Deacons Corporation is presented below. At year-end, the following additional information is available: 1. The balance of Prepaid Rent,...
-
Your mother just received a $250,000 inheritance. If she invests her money in a diversified equity portfolio returning 8 percent per year, approximately how long will it take her to become a...
-
Return to a risk-free rate of 3.0 percent. Now suppose the government announces substantial changes to the tax code. The uncertainty causes investors to become more risk averse, and the market risk...
-
The company Ogivac Inc. has the possibility of buying a property costing 70 000 $ and which is depreciable, for tax purposes, at the declining rate of 30%. The value expected residual at the...
-
Explain the capital markets both domestic and foreign used by firms to obtain long-term financing. Explain the capital markets both domestic and international. What are they and how do they function...
-
Using the data below, your task is to negotiate the purchase price of the Target property with another team. All comps are within a 2 mile radius of the Target Property and represent transactions...
-
Evaluate the integral by changing to spherical coordinates. 1 T-x 2-*-? dz dy dx
-
Suppose that a business sells 6-month subscriptions to its monthly magazine. On January 1, the company receives a total of $600 for 10 subscriptions. To record this transaction, the company debits...
-
Solve for the hydrogen-ion concentration in solutions of acetic acid with stoichiometric molarities equal to 0.00100 mol l 1 . Use the method of successive approximations.
-
Verify the prediction of the ideal gas equation of state given in the previous example.
-
Substitute the value of the molar volume obtained in the previous example and the given temperature into the Dieterici equation of state to calculate the pressure. Compare the calculated pressure...
-
What is the expected rate of return on a project that requires an investment of $106 today and generates cash inflows of $7, $17 and $122 in each of the next 3 years?
-
A trader opens a new position by writing two put option contracts. Each contract is on 100 shares of Exxon Mobil common stock. The option premium is $6.06, the strike price is $50, and the stock...
-
On a particular day, there were 300 stocks that advanced on the NYSE and 800 that declined. The volume in advancing issues was 1000 and the volume in declining issues was 3000. What is the common...

Study smarter with the SolutionInn App


