How do the results shown in Figure 20.10 differ from the predictions of the Bohr model of
Question:
Figure 20.10
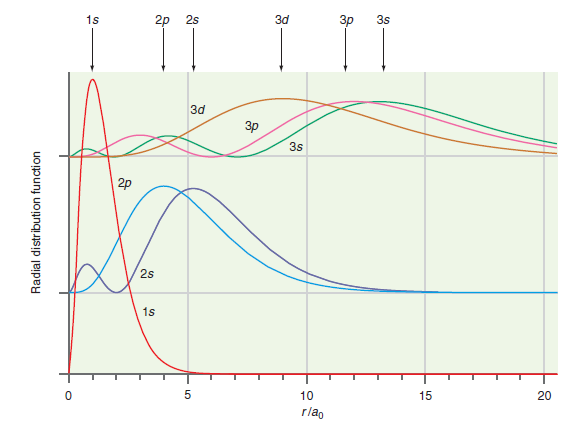
Transcribed Image Text:
1s 2p 2s Зd Зр 3s Зd Зр 3s 2p 2s 1s 10 15 20 rlag Radial distribution function
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
The Bohr model predicts that the electron ...View the full answer

Answered By

Sultan Ghulam Dastgir
The following are details of my Areas of Effectiveness English Language Proficiency, Organization Behavior , consumer Behavior and Marketing, Communication, Applied Statistics, Research Methods , Cognitive & Affective Processes, Cognitive & Affective Processes, Data Analysis in Research, Human Resources Management ,Research Project,
Social Psychology, Personality Psychology, Introduction to Applied Areas of Psychology,
Behavioral Neurosdence , Historical and Contemporary Issues in Psychology, Measurement in Psychology, experimental Psychology,
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Discuss whether the results shown in Figure 17.7 are consistent with local realism. Figure 17.7 300 250 200 150 100 50 -8 -6 -4 -2 4 6. 8. Detector 2 position/mm Coincidence counts
-
A certain compound has a molecular weight of 83 and contains nitrogen. Its infrared spectrum contains a moderately strong peak at 2270 cm-1. Its 1H and 13C NMR spectra are shown in Figure 20.10. What...
-
(a) What is the role of the porous glass disc shown in Figure 20.4? (b) Why do NO3- ions migrate into the anode half-cell as the voltaic cell shown in Figure 20.5 operates?
-
Your geothermal heat pump is used to heat your house in the winter. The heat pump is operated as a vapor-compression cycle. The design is such that there must be a 10 degree temperature difference...
-
Verify that each x-value is a solution of the equation. 1. tan x - 3 = 0 (a) x = / 3 (b) x = 4 / 3 2. sec x -2 = 0 (a) x = / 3 (b) x = 5 / 3 3. 3 tan2 2x - 1 = 0 (a) x = / 12 (b) x = 5 / 12 4. 2...
-
A control chart for the number of nonconforming piston rings is maintained on a forging process with np = 16.0. A sample of size 100 is taken each day and analyzed. (a) What is the probability that a...
-
A company that has 200 employees chooses a committee of 5 to represent employee retirement issues. When the committee is formed, none of the 56 minority employees are selected. (a) Use technology to...
-
For the year ended December 31, 2012, the job cost sheets of Dosey Company contained the following data. Other data: 1. Raw materials inventory totaled $20,000 on January 1. During the year, $100,000...
-
Waiting in the South Bend, Indiana jail for the trial on the properties that nana (grandma) purchased in New York for the trial that was upcoming and to be held in the Northern District of Indiana....
-
A new instrument capable of performing 40,000 tests per year has a purchase price of $15,000,000. The installation will cost 10% of the purchase price. The manufacturer covers maintenance costs for...
-
Why are the total energy eigenfunctions for the H atom not eigenfunctions of the kinetic energy?
-
What effect does the centrifugal potential have in determining the maximum in the radial function for the 3s, 3p, and 3d orbitals?
-
How would you prioritize the considerations in menu planning for your restaurant?
-
Write a short-note on bio-fuels.
-
Give detailed classification of fuel.
-
Discuss the use of fuels and their classifications.
-
Discuss the function of condensers.
-
What do you mean by calorific value? Explain the experimental setup to measure the calorific value of a liquid fuel.
-
Use a trigonometric identity to show that the derivatives of the inverse cotangent and inverse cosecant differ from the derivatives of the inverse tangent and inverse secant, respectively, by a...
-
Hotel Majestic is interested in estimating fixed and variable costs so that the company can make more accurate projections of costs and profit. The hotel is in a resort area that is particularly busy...
-
A correlation for methane solubility in seawater is given by the equation where is volume of gas in mL at STP per unit volume (mL) of water when the partial pressure of methane is 760 mm Hg, T is...
-
A liquid mixture containing 50 mole% propane, 30% n-butane, and 20% isobutane is stored in a rigid container at 77F. The container has a maximum allowable working pressure of 400 psig. The head space...
-
A liquid mixture contains N components (N may be any number from 2 to 10) at pressure Pmm Hg. The mole fraction of the ith component is xi(i = 1; 2; ... ;N), and the vapor pressure of that component...
-
What strategies might you use to implement the personal change needed to support organizational change? What is the relevance of unity consciousness in organizational leadership, communications, and...
-
Recognizing the literature (and its limitations) for conveying policing organization, management, and change - what management/organizational changes are needed in policing today? Why? How do we set...
-
How do organizations effectively navigate the complexities of organizational change, integrating advanced change management methodologies to mitigate resistance and foster sustainable transformation?

Study smarter with the SolutionInn App


