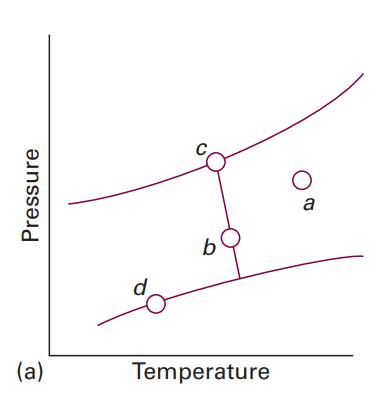
How many phases are present at each of the points marked in Fig. 4.1a? Data in Fig.
Question:
How many phases are present at each of the points marked in Fig. 4.1a?
Data in Fig. 4.1a?
Transcribed Image Text:
Pressure (a) d b Temperature O a
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
There are 3 phases present at any point marked in Fig 41a i...View the full answer

Answered By

Enock Oduor
I am a chemist by profession, i coach high school students with their homework, i also do more research during my free time, i attend educational and science fair seminars where i meet students and do some projects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
Mr. Lee, clinic director claims that through new procedures he has reduced patient waiting time from present 55 minutes. Before implementing his procedures nationwide, the clinic owner, Ms. Groetken,...
-
Each of the points listed next represents an internal control that may be implemented within a companys accounting information system to reduce various risks. For each point, identify the appropriate...
-
Consider the following phase diagram. What phases are present at points A through H? Identify the triple point, normal boiling point, normal freezing point, and critical point. Which phase is denser,...
-
Carey Company is borrowing $200,000 for one year at 12 percent from Second Intrastate Bank. The bank requires a 20 percent compensating balance. What is the effective rate of interest? What would the...
-
How do firms record prior- period adjustments? .
-
What is meant by "audit quality," and what factors influence it?
-
Consider the PDE problem where \ (a, b\), and \(\omega\) are constants. Write the full finite difference discretization of the problem on a uniform grid. The discretization must be of the first order...
-
Donna Shader, manager of the Winter Park Hotel, is considering how to restructure the front desk to reach an optimum level of staff efficiency and guest service. At present, the hotel has five clerks...
-
1. (CASH FLOW FOR OPERATING ACTIVITIES USING THE DIRECT METHOD) Perval's income statement for last year appears below: Perval Company Income statement Sales P 100,00 0 Cost of goods sold (60,00 P 0)...
-
Pan Company issued 960,000 shares of $10 par common stock with a fair value of $20,400,000 for all the voting common stock of Set Company. In addition, Pan incurred the following costs: Legal fees to...
-
The protein lysozyme unfolds at a transition temperature of 75.5 C and the standard enthalpy of transition is 509 kJmol 1 . Calculate the entropy of unfolding of lysozyme at 25.0 C, given that the...
-
Calculate the change in entropy of the system when 10.0 g of ice at 10.0 C is converted into water vapour at 115.0 C and at a constant pressure of 1bar. The constant-pressure molar heat capacity of H...
-
Rewrite the MyLinkedList class without using header and tail nodes and describe the differences between the class and the class provided in Section 3.5.
-
Mr. Jernigan owns a piece of land on which he grows corn. Corn production annually requires $2,000 in seed, $3,000 in fertilizer, and $6,000 in pesticides. Mr. Jernigan uses his own labor to grow the...
-
The covariance between the market return on the shares in the company COV and the return on the market portfolio is 0.02. The standard deviation of the return on the shares in COV is 20%. The...
-
For this assignment, you are to make a linguistic autobiography research paper that connects your experience as a user of one or more American language to concepts covered throughout this course....
-
Define yield to maturity. How do changes in the market YTM affect the value of a bond?
-
Lana purchased a powerful laptop computer for $3,000. She earned $2557 mining bitcoins before selling it a year later for $1601. What was her holding period return?
-
Refer to the DaisyMate data set. Assume the company uses a standard cost accounting system. In Daisy Mate Data set Direct materials (resin) .......................................... 5 pounds per pot...
-
Smthe Co. makes furniture. The following data are taken from its production plans for the year. Required: 1. Determine the hazardous waste disposal cost per unit for chairs and for tables if costs...
-
Hexane and perfluorohexane show partial miscibility below 22.70C. The critical concentration at the upper critical temperature is x = 0.355, where x is the mole fraction of C 6 F 14 . At 22.0C the...
-
Some polymers can form liquid crystal mesophases with unusual physical properties. For example, liquid crystalline Kevlar (3) is strong enough to be the material of choice for bulletproof vests and...
-
Refer to the information in Exercise 6.15(b) and sketch the cooling curves for liquid mixtures in which x(B 2 H 6 ) is (a) 0.10, (b) 0.30, (c) 0.50, (d) 0.80, and (e) 0.95. Data in Exercise 6.15(b)...
-
In addition to calculating earnings, deductions and employer obligations arising from payroll, payroll administrators must ensure that all liabilities resulting from the payroll process are paid on...
-
2. Answer chapter 5 Study Questions and Problems in the ASP for question numbers 1, 2, 3, 4, 5,6,7,8,9,11,12,13,16 on page 83.
-
Potential output is the level of output when all _ _ _ _ _ _ _ _ are in equilibrium.

Study smarter with the SolutionInn App


