Identify the molecular orbitals for F 2 in the images shown here in terms of the two
Question:
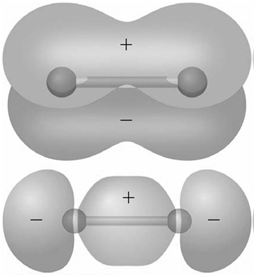
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
The top image is of symmetry and has no ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Draw the molecular orbitals for the cyclopropenyl case. (Since there are three p orbitals, there must be three MOs: one all-bonding MO and one degenerate pair of MOs.) (b) Draw an energy diagram...
-
Evaluate the Klopp case in terms of those two criteria.
-
Images of molecular orbitals for LiH calculated using the minimal basis set are shown here. In these images, the smaller atom is H. The H1s AO has a lower energy than the Li2s AO. The energy of the...
-
9:49 X Bank Reconciliation Assignment 1 PDF 150 KB Assignment #1 - Bank Reconciliation This assignment has 33 total marks. This assignment is worth 5% of your final grade. This assignment is due...
-
A police car with its spotlight on is parked 20 meters from a warehouse. Consider and x as shown in the figure. (a) Write θ as a function of x. (b) Find θ when x = 5 meters...
-
Is there evidence consistent with gender discrimination in training level? To answer this, proceed as follows: a. Create a table of counts for the two qualitative variables gender and training level....
-
Consider two countries, A and B, with the technologies given by case 3 in Exercise 1. data from case 3 in exercise 1 Suppose that the wage rate in A, WA, equals \($9\) per hour; then, for mutually...
-
Transaction data for Sanculi Real Estate Agency are presented in E2-4. In E2-4, the following information relates to Sanculi Real Estate Agency. Oct. 1 Alan Sanculi begins business as a real estate...
-
During a class discussion, Jamie said that she changes the form of notetaking she uses depending on the complexity of the material she is reading. The teacher praised her methods for adjusting the...
-
a. Using revenue management, determine how many seats should be reserved for premium ticket holders. b. Considering your answer to part (a) and the possible outcomes listed above, how much total...
-
If there is a node in u , is the electron in this wave function really delocalized? How does it get from one side of the node to the other?
-
By considering each term in And Explain why the values of J and K are positive for H + 2 . e? |e, dr - j (, Hs, dt J = [@n| dt 4TEQ7B.
-
Incaviglia Company sold 400 copy-making machines in 2014 for $3,000 apiece, together with a one-year warranty. Maintenance on each machine during the warranty period averages $330. Instructions (a)...
-
You stand in your yard and hold a battery-powered buzzer that is emitting a frequency of \(560 \mathrm{~Hz}\) as a friend stands next to you. (a) What frequency does your friend hear if you throw the...
-
Two airplanes simulating a dogfight are headed right for each other, plane A moving at \(285 \mathrm{~km} / \mathrm{h}\) and plane B moving at \(295 \mathrm{~km} / \mathrm{h}\). The electronic...
-
While you are driving on a straight road at \(97 \mathrm{~km} / \mathrm{h}\), a police car traveling in the opposite direction comes toward you with the siren blaring, and the frequency you hear is...
-
A car traveling west at \(90 \mathrm{~km} / \mathrm{h}\) along a road running parallel to a railroad track approaches a train traveling east at \(65 \mathrm{~km} / \mathrm{h}\). If the train's...
-
Two violin strings, each having a linear mass density of \(0.0014 \mathrm{~kg} / \mathrm{m}\) and under the same \(100-\mathrm{N}\) tension, have the same fundamental frequency of \(660...
-
Fill in the blank with an appropriate word, phrase, or symbol(s). Jeffersons method, Websters method, and Adams method require using a(n) __________ quota.
-
a. What is meant by the term tax haven? b. What are the desired characteristics for a country if it expects to be used as a tax haven? c. What are the advantages leading an MNE to use a tax haven...
-
The CH 3 Cl molecule belongs to the point group C 3v . List the symmetry elements of the group and locate them in the molecule.
-
Show that, for a perfect gas, (U/S) V = T and (U/V) S = p.
-
Sketch the form of the 19 F-NMR spectra of a natural sample of tetrafluoroborate ions, BF 4 , allowing for the relative abundances of 10 BF 4 and 11 BF 4 .
-
Total Labor- Square Feet of Department Hours Space Occupied Number of Employees Machine- Hours Direct Labor- Hours Personnel 16,000 12,400 22 Custodial Services 8,200 3,100 49 Maintenance 14,300...
-
6. Given the Venn Diagram, find (AUB)NC A B a W b Y N X C C
-
Prepare one-page description of the company and a one-page description of the project. In the project description, please list all the key stakeholders who will attend the project kickoff meeting....

Study smarter with the SolutionInn App


