Identify the molecular orbitals for F 2 in the images shown here in terms of the two
Question:
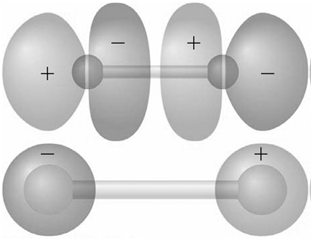
Transcribed Image Text:
+ +
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
The top image has symmetry the AOs are out of ...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Draw the molecular orbitals for the cyclopropenyl case. (Since there are three p orbitals, there must be three MOs: one all-bonding MO and one degenerate pair of MOs.) (b) Draw an energy diagram...
-
Evaluate the Klopp case in terms of those two criteria.
-
Images of molecular orbitals for LiH calculated using the minimal basis set are shown here. In these images, the smaller atom is H. The H1s AO has a lower energy than the Li2s AO. The energy of the...
-
One of Red Clay's employee handbook sections covers employee monitoring. An employee filed a complaint with the HR department stating he opposes this monitoring and chooses to opt out. What legal...
-
Different types of granular substances naturally settle at different angles when stored in cone-shaped piles. This angle θ is called the angle of repose (see figure). When rock salt is...
-
Glentech Manufacturing is considering the purchase of an automated parts handler for the assembly and test area of its Phoenix, Arizona, plant. The handler will cost $ 250,000 to purchase plus $...
-
According to Figure 1.2, intraEuropean Union trade accounts for a huge proportion of EU trade. What factor or factors might account for this fact? Asiab exports: $19.9 billion to U.S. & Canada ($4.9)...
-
David Rodriguez owns a chain of travel goods stores, Rodriguez Travel Goods. Last year, his sales staff sold 25,000 suitcases at an average sales price of $150. Variable expenses were 60% of sales...
-
Hudson Company reports the following contribution margin income statement. HUDSON COMPANY Contribution Margin Income Statement For Year Ended December 31 Sales (11,200 units at $280 each) Variable...
-
QSR magazine reports on the largest quick serve and fast casual restaurants in the United States. Do the various market segments (burger, chicken, sandwich, and pizza) differ in their mean sales per...
-
Explain why the nodal structures of the 1 g MOs in H 2 and F 2 differ.
-
In discussing Figure 23.2, the following statement is made: Interchanging red and blue does not generate a different MO. Justify this statement. Figure 23.2 H1s His H2 Energy
-
The amount of subjectivity involved in establishing fair value estimates can be complex for management responsible for making the fair value measurements and disclosures contained in financial...
-
Henredon purchases a high-precision programmable router for shaping furniture components for \($190,000.\) It is expected to last 12 years and have a salvage value of \($5,000.\) Henredon will borrow...
-
Resonances of the ear canal lead to increased sensitivity of hearing, as we've seen. Dogs have a much longer ear canal\(5.2 \mathrm{~cm}\)-than humans. What are the two lowest frequencies at which...
-
A dog can provide sufficient power to pull a sled with a 60 N force at a steady 2.0 m/s. Suppose the dog is hitched to a different sled that requires 120 N to move at a constant speed. How fast can...
-
At December 31, 2025, Carlson Enterprises had accounts receivable of 750,000. On January 1, 2025, Allowance for Doubtful Accounts had a credit balance of 18,000. During 2025, 30,000 of uncollectible...
-
A \(4.0-\mathrm{kg}\) block and a \(2.0-\mathrm{kg}\) block are connected to opposite ends of a relaxed spring of spring constant \(300 \mathrm{~N} / \mathrm{m}\). The blocks are pushed toward each...
-
Fill in the blank with an appropriate word, phrase, or symbol(s). If a candidate is the winner of an election and in a second election one or more of the other candidates is removed, the previous...
-
The Dow Jones Industrial Average reached a high of $ 7801.63 on December 29, 1997. Recall from Example 18.4 that it reached a high of $ 1003 on November 14, 1972. The Consumer Price Index for...
-
Discuss the origins of diagonal and cross peaks in the COSY spectrum of an AX system.
-
Calculate the frequency separation of the nuclear spin levels of a 13 C nucleus in a magnetic field of 14.4 T given that the magnetogyric ratio is 6.73 10 7 T 1 s 1 .
-
33 S has a nuclear spin of 3/2 and a nuclear g-factor of 0.4289. Calculate the energies of the nuclear spin states in a magnetic field of 7.500 T.
-
The Pew Internet and American Life Project reported on the widespread use of the Internet to search for information regarding experimental treatments or medicines. From telephone interviews of 1005...
-
write the differences between Microprocessors and Microcomputers. compare between them and draw the block diagram for each one. B) define the interface and mention it's types with brief description....
-
Write a program that prompts the user for the constitutive parameters (relative permittivity, relative permeability, and conductivity) in medium 1 and medium 2 separated by a planar surface. You are...

Study smarter with the SolutionInn App


