Name the alkane: CHs CHy-CH-CH -HS CH2-CH
Question:
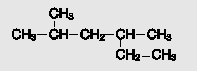
Transcribed Image Text:
CHs CHy-CH-CH ҫн-сHS CH2-CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (8 reviews)
2 4dimethylhexan...View the full answer

Answered By

Tamil Elakkiya Rajendran
I'm currently involved in the research in the field of Biothermodynamics, Metabolic pathway analysis and computational Biology. I always prefer to share my knowledge whatever I have learnt through my degree whenever time permits.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
All the parts of this problem refer to the alkane having the carbon skeleton shown. (a) What is the molecular formula of this alkane? (b) What is its IUPAC name? (c) How many methyl groups are...
-
Refer to Table 2.4 as needed to answer the following questions: (a) Octacosane has been found to be present in a certain fossil plant. Write a condensed structural formula for octacosane. (b) What is...
-
Name the class to which each of the compounds used in this experiment belongs (eg. alcohol, alkane, ester, etc.)
-
The AFL-CIO has undertaken a study of the yearly salaries (in thousands of dollars) of 30 administrative assistants. The organization wants to predict salaries from several other variables. The...
-
For each of the following overhead costs, suggest one method of apportioning cost to cost centre: (a) Employees' holiday pay; (b) Depreciation of floor-polishing machines used in all production...
-
Does the valuation of an asset-backed security require the use of Monte Carlo simulation?
-
Why is the finite element approach presented in this chapter called the displacement method?
-
Techno Designs produces head covers for golf clubs. The company expects to generate a profit next year. It anticipates fixed manufacturing costs of $126,500 and fixed general and administrative...
-
10. Horace uses the cash basis to account for revenue and expenses in his business. He has the following business revenue and expenses during the year: Gross receipts Business expenses Customer...
-
Evaluate the following pure-yield pickup swap: You currently hold a 20-year, AA-rated, 9.0 percent coupon bond priced to yield 11.0 percent. As a swap candidate, you are considering a 20-year,...
-
A papyrus scroll was buried with King Tutankhamen 3330 years ago. The half-life of carbon-14 is 5730 years. The papyrus scroll sample had an initial decay of 15.3 disintegrations per minute per gram...
-
Name the alkene: CHs- CH-CH-- CH2-CHa
-
Many people are less concerned with the absolute size of the government debt than they are about its size relative to GDP. Such people would not worry about the size of government deficits if the...
-
All but one of the following is reported in a retained earnings statement. The exception is: a. share dividends. b. net income and net loss. c. sales revenue. d. cash dividends.
-
XYZ Ltd. sells 100 of its 5 par value treasury shares at 13 per share. If the cost of acquiring the shares was 10 per share, the entry for the sale should include credits to: a. Treasury Shares 1,000...
-
The following data are available for Orange Peels Ltd. Net cash provided by investing activities is: a. NT$1,200,000. b. NT$1,300,000. c. NT$1,500,000. d. NT$1,900,000. Sale of land NT$1,000,000 Sale...
-
A vertical spring on which is hung a block of mass \(m_{1}\) oscillates with frequency \(f\). With an additional block of mass \(m_{2} eq m_{1}\) added to the spring, the frequency is \(f / 2\). What...
-
Create a six-digit coding system which would allow you to classify all the items of expenditure you make in a year. ( You will need to write down the items of expenditure first of all and then look...
-
Graph each polynomial function. (x) = x 3 - 5x 2 + 3x + 9
-
For the following exercises, find the area of the triangle. Round to the nearest hundredth. 22 50 36
-
Ethylene glycol (antifreeze) has a density of 1.11 g/cm 3 . a. What is the mass in g of 417 mL of ethylene glycol? b. What is the volume in L of 4.1 kg of ethylene glycol?
-
A supposedly gold nugget displaces 19.3 mL of water and has a mass of 371 g. Could the nugget be made of gold?
-
Glycerol is a syrupy liquid often used in cosmetics and soaps. A 3.25 L sample of pure glycerol has a mass of 4.10 * 10 3 g. What is the density of glycerol in g/cm 3 ?
-
The Step Company has the following Information for the year just ended: Sales in units Budget 15,000 Actual 14,000 Sales $150,000 $147,000 Less: Variable Expenses 90,000 82,600 Contribution Margin $...
-
Question 2 (2 marks) The year level coordinators for Years 7 to 11 at the school are Amy (A), Brian (B), Claire (C), Daisy (D) and Ellie (E). A faulty telephone system means that some of these...
-
On a certain farm alfalfa yields have been 4.3, 7.2, 5.6, and 3.6 tons per acre in the last 4 years. What is the expected value for the farm's alfalfa yield, if each past result is given equal weight...

Study smarter with the SolutionInn App


