Show that the expression (U/V) T = T(P/T)V P can be written in the form 2 /
Question:
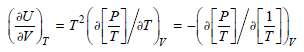
Transcribed Image Text:
т2 /т av ३
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
We now change the differentiation to the ...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show that for nonmagnetic media, the reflection coefficient k can be written in the form. tan(e,- 0,) tan(0; + 0,)
-
Show that the reflection coefficient can be written in the form. sin(e - sin(0, + 0;)
-
Show that the electric field of a ("pure") dipole (Eq. 3.103) can be written in the coordinate-free form (2 cos e f + sin ). Edip (r, A) (3.103) 40r3
-
WESTON ENTERPRISES 2019 and 2020 Partial Balance Sheets Assets 2019 2020 Current assets $1,193 $1272 Net fixed assets 5,722 6,023 WESTON ENTERPRISES 2020 Income Statement Sales Costs Depreciation...
-
What is the term for a firm that holds such market power that it can limit rivals from competing in the market?
-
Beginning with Eq. (14.50), prove Eq. (14.52), and derive the chiral decompositions given for \(\bar{\psi} \psi, \bar{\psi} \gamma^{\mu} \psi\), and \(\bar{\psi} \gamma^{\mu} \gamma^{5} \psi\)...
-
Architecture is defined from two perspectives: the product itself and the development of the product. Compare the two definitions.
-
On August 1, 2013, Sietens Corporation had the following account balances: Raw Material Inventory (both direct and indirect) ..... $ 72,000 Work in Process Inventory .............. 108,000 Finished...
-
Shep Company's records show the following information for the current year: Total assets Total liabilities Beginning of year $ 57,200 End of year $ 89,000 $ 25,600 $ 38,600 Determine net income...
-
A soft drink bottling company is interested in controlling its filling operation. Random samples of size 4 are selected and the fill weight is recorded. Table 7-17 shows the data for 24 samples. The...
-
True or False. Under the PPACA, Americans are free to decide for themselves whether or not they should have health insurance coverage.
-
A sample containing 2.50 mol of an ideal gas at 325 K is expanded from an initial volume of 10.5 L to a final volume of 60.0 L. Calculate G and A for this process for a. An isothermal reversible path...
-
Determine the voltage at each point with respect to ground in Figure 478. 15 V A R 560 R 560 FIGURE 4-78 B R3 100 C R4 1.0 D R 100
-
What is the difference between weak-tie and strong-tie support? Where do the people in your life fit? When and why would you seek support from someone instead of someone else?
-
How does consulting with children encourage involvement? Give at least four examples.
-
16dx Evaluate I = 9- 4x2
-
What nuanced methodologies exist for evaluating and selecting high-performing mutual funds or exchange-traded funds (ETFs) that align with an investor's specific investment goals and risk tolerance...
-
The price of a UK 10-year gilt is quoted at 10713/32 It has a face value of 100, and yields coupons at a rate of 4.25%, that it pays semi-annually. What is the price of the bond today? Comment on...
-
A board game uses the deck of 20 cards shown. Two cards are selected at random from this deck. Determine the probability of the following (a) With replacement. (b) Without replacement. They both show...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
The standard enthalpy of combustion of solid urea (CO (NH2)2) is -632 kl mol-1 at 298 K and its standard molar entropy is 104.60 J K-1 mol-1, Calculate the standard Gibbs energy of formation of urea...
-
Calculate the change in the entropies of the system and the surroundings, and the total change in entropy, when the volume of a sample of argon gas of mass 21 gat 298 K and 1.50 bar increases from...
-
The use of supercritical fluids as mobile phases in SFC depends on their properties as non polar solvents. The solubility parameter, , is defined as (Ucohesive/Vm)1/2, where Ucohesive is the cohesive...
-
I am running for Vice President of Legislative Affairs at my University as part of the Student Government Association. I am need of ideas!!! Keep this in mind. My platform is centered around pushing...
-
What are some ways that synergistic communication can positively affect organizational communication? Furthermore, how does SC relate to the achieving of (or the not achieving of) the overall goals...
-
Choose an organization's website. Provide the name of the company and the URL address at the top of your answer. i) Analyze the website using THREE (3) relevant corporate communication concepts. (9%)...

Study smarter with the SolutionInn App


