Starting from the expression C p C V = T(p/T) V (V/T) p , use the
Question:
Starting from the expression Cp − CV = T(∂p/∂T)V(∂V/∂T)p, use the appropriate relations between partial derivatives to show that
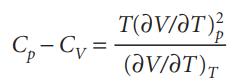
Evaluate Cp − CV for a perfect gas.
Transcribed Image Text:
C-C= ᎢᏧᏙ/Ꭲ ) V/T).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Answer The partial derivatives of PV with respect to pressure are zero heat does not change pressure ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show that for an ideal gas cp = cv + R u.
-
Starting from the expression for the MOSFET unity-gain frequency, fT=gm/2*pi*(Cgs+Cgd) and making the approximation that Cgs>>Cgd and that the overlap component of Cgs is negligibly small, show...
-
Starting from the expression of the total energy carried by the lattice vibrations in Eq. (6.60), show that the heat capacity C v = (dE/dT) v can be written as: T C, = 9Nk, Op (et 1)2
-
My division had another great year last year. We all worked hard, and the results were there. But again we got no reward for our hard work. It's very frustrating. - Division Manager, General Products...
-
What are liabilities? Define current liabilities and long-term liabilities.
-
Find two circle graphs in newspapers, in magazines, or on the Internet whose data are not given in percents. Redraw the graphs and label them with percents.
-
The number of degrees of freedom for a system prepared by partially decomposing \(\mathrm{NH}_{4} \mathrm{Cl}\) into an evacuated space is (a) 0 (b) 1 (c) 2 (d) 3 .
-
Richard Schewe and others placed personal property in a building occupied by Winnebago County Fair Association, Inc. Prior to placing their property in the building, they signed a Storage Rental...
-
Laurel Enterprises expects earnings next year of $4.31 per share and has a 50% retention rate, which it plans to keep constant. Its equity cost of capital is 9%, which is also its expected return on...
-
Cecilias Boutique wants several lists of salesperson data. Design a flowchart or pseudocode for the following: a. A program that accepts one salespersons ID number, number of items sold in the last...
-
The standard enthalpy of decomposition of the yellow complex H 3 NSO 2 into NH 3 and SO 2 is +40 kJ mol 1 . Calculate the standard enthalpy of formation of H 3 NSO 2 .
-
Show that the following functions have exact differentials: (a) X 2 y + 3y 2 , (b) X cos xy, (c) X 3 y 2 , (d) T(t + e s ) + s.
-
Your class observed several different objects in motion along different lines. Figure P2.5 shows some of the graphs other students made of the events. They have labeled the horizontal axis "time" and...
-
The credit manager of Montour Fuel has gathered the following information about the company's accounts receivable and credit losses during the current year: Net credit sales for the year $ 7 , 5 0 0...
-
Sub Station and Planet Sub reported the following selected financial data ( $ in thousands ) . Sub Station s business strategy is to sell the best tasting sandwich with the highest quality...
-
How truly do drive elements and social impact instruments, for example, social personality hypothesis and gathering polarization, deeply mold hierarchical way of behaving and decision-production at...
-
Harbert, Incorporated had a beginning balance of $12,000 in its Accounts Receivable account. The ending balance of Accounts Receivable was $10,500. During the period, Harbert collects $72,000 of its...
-
Milden Company is a merchandiser that plans to sell 4 1 , 0 0 0 units during the next quarter at a selling price of $ 5 9 per unit. The company also gathered the following cost estimates for the next...
-
Give a new proof of Theorem PSPHS replacing applications of Theorem SLSLC with matrix-vector products (Theorem SLEMM).
-
For the vector whose polar components are (Vr = 1, Vθ = 0), compute in polars all components of the second covariant derivative Vα;μ;ν. To find...
-
A light atom rotating around a heavy atom to which it is bonded is described by a wavefunction of the form ()=e im with 02 and m an integer. Show that the m=+1 and m=+2 wavefunctions are orthogonal.
-
For a black body, the temperature and the wavelength of emission maximum, max , are related by Wiens law, max T = 1/5c 2 , where c 2 =hc/k (see Problem 7A.3). Values of max from a small pinhole in...
-
Describe the properties of wave packets in terms of the Heisenberg uncertainty principle.
-
Compare and contrast digital evidence and non-digital evidence. Provide three examples of how they are similar and three examples of how they are different. You must use the provided course materials...
-
In May of 2015, Mr. Miradi made an oral agreement with Mr. Miondoko for the sale of a piece of land situate in Nyeri County Land title No. Gakawa/Kahurura Block III/Mwichwiri/823 for a price of KShs....
-
and QB- Two identical point charges (+2.27 10-9 C) are fixed in place, separated by 0.460 m (see the figure). Find (a) the electric field and (b) the electric potential at the midpoint of the line...

Study smarter with the SolutionInn App


