Taking as references pure liquid sulfuric acid at 77F and pure liquid water at 32F and without
Question:
Taking as references pure liquid sulfuric acid at 77°F and pure liquid water at 32°F and without using Figure 8.5-1, calculate Ĥ(Btu/lbm) for each of the following substances. For each substance, also report the value you would read from Figure 8.5-1.
(a) H2O (l, 120°F)
(b) H2SO4 (l, 200°F)
(c) 60 wt% H2SO4 (aq, 200°F)
Figure 8.5-1
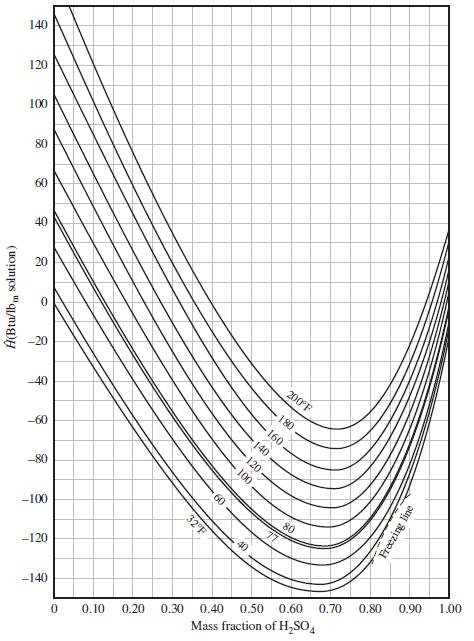
Transcribed Image Text:
140 120 100 80 60 40 20 -20 200°F -40 180 -60 -80 60 32°F 80 -100 40 -120 0.90 1.00 0.80 -140 0.70 0.50 0.60 0.40 0.30 0.10 0.20 Mass fraction of H,SO, A(Btu/lb, solution) Freezing line
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
To calculate HBtulbm we can use the following formula H H H0 where H is the enthalpy of the substanc...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-1119498759
4th edition
Authors: Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Question Posted:
Students also viewed these Sciences questions
-
A closed, insulated container is charged with a stoichiometric ratio of oxygen and hydrogen at 77 F and 20 lbf/in 2. After combustion, liquid water at 77 F is sprayed in such that the final...
-
Water is added to pure sulfuric acid in a well-insulated flask initially at 25C and 1 atm to produce a 4.00-molar sulfuric acid solution (SG = 1.23 1). The final temperature of the product solution...
-
One alternative to using petroleum or natural gas as fuels is ethanol (C2H5OH), which is commonly produced from grain by fermentation. Consider a combustion process in which liquid ethanol is burned...
-
Does the performance of kinross gold cooperation affect the performance S&P/TSX Composite index? Explain why or why not using this graph in brief Kinross Gold Corporation (K.TO) Toronto - Toronto...
-
List the sources and uses of secondary data. What are the benefits of secondary data? What are the limitations?
-
How can speakers adapt their message and delivery to the needs of the audience? Why should they?
-
Identify a minimum of five kinds of motions some- times filed in divorce actions while the litigation is pending.
-
Gemstone Quarry is trying to decide whether to invest in a new material-handling system. The current system (which is old and completely paid for) has an annual maintenance cost of $10,000 and costs...
-
In the year to 30 September 2022, an advertising agency declares in interim ordinary dividend of 7.4C per shares and a final ordinary dividend of 8.6C per share. assuming an ex-dividend share price...
-
Hunter Company is developing its annual financial statements at December 31, 2017. The statements are complete except for the statement of cash flows. The completed comparative balance sheets and...
-
Aqueous sulfuric acid solutions containing 15.0 wt% H 2 SO 4 and 80.0 wt% H 2 SO 4 are mixed to form a 30.0 wt% product solution. The 15% solution was in a laboratory in which the temperature was...
-
The standard heat of the reaction is H r = 904:7 kJ. (a) Briefly explain what that means. Your explanation may take the form When ______ (specify quantities of reactant species and their physical...
-
Temporary employees are being used with increasing regularity throughout the world. In France, one in five workers is on a temporary or part-time contract, and in the UK more than 25 percent of the...
-
You are considering how to invest part of your retirement savings. You have decided to put $300,000 into three stocks: 60% of the money in GoldFinger (currently $23/share), 30% of the money in...
-
You own three stocks: 600 shares of Apple Computer, 10,000 shares of Cisco Systems, and 5000 shares of Colgate-Palmolive. The current share prices and expected returns of Apple, Cisco, and...
-
Consider two local banks. Bank A has 76 loans outstanding, each for $1 million, that it expects will be repaid today. Each loan has a 6% probability of default, in which case the bank is not repaid...
-
Consider the following two, completely separate, economies. The expected return and volatility of all stocks in both economies is the same. In the first economy, all stocks move togetherin good times...
-
Consider an economy with two types of firms, S and I. S firms all move together. I firms move independently. For both types of firms, there is a 70% probability that the firms will have a 7% return...
-
A narrow beam of light from a laser travels through air (n = 1.00) and strikes point A on the surface of the water (n = 1.33) in a lake. The angle of incidence is 55. The depth of the lake is 3.0 m....
-
Which of the following gives the range of y = 4 - 2 -x ? (A) (- , ) (B) (- , 4) (C) [- 4, ) (D) (- , 4] (E) All reals
-
Deduce the structure of a compound with molecular formula C 9 H 12 that produces the following 1 H NMR spectrum: Proton NMR Chemical Shift (ppm)
-
Deduce the structure of a compound with molecular formula C 9 H 10 O 2 that produces the following 1 H NMR spectrum and 13 C NMR spectrum: Proton NMR 10 Chemical Shift (ppm) Carbon NMR - 128.4 128.8-...
-
Propose the structure of a compound consistent with the following data: (a) C 5 H 10 O, broadband-decoupled 13 C NMR: 7.1, 34.6, 210.5 (b) C 6 H 10 O, broadband-decoupled 13 C NMR: 70.8, 116.2, 134.8
-
How do you measure team compliance and competence of the roles allocated within the budget/financial plans?
-
How do demographic shifts, including changes in population dynamics, migration patterns, and urbanization, shape the landscape of social change at both local and global scales?
-
How do emergent macro-level phenomena, such as globalization and technological advancements, catalyze social change across diverse cultural landscapes?

Study smarter with the SolutionInn App


