The amino acid glycine dimerizes to form the dipeptide glycylglycine according to the reaction 2Glycine(s) Glycylglycine(s) +
Question:
2Glycine(s) †’ Glycylglycine(s) + H2O(l)
Calculate ΔS, ΔSsurroundings , and ΔSuniverse at T = 298 K. Useful thermodynamic data follow:
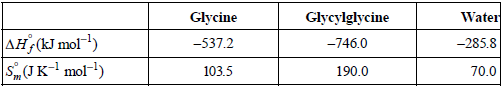
Transcribed Image Text:
Glycylglycine Water -285.8 Glycine -537.2 AH (kJ mol) SJK- mol-!) -746.0 103.5 190.0 70.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
ASR 2 1035JK mol 1900 J K mol700 JK mol 5...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The diffusion constant for the amino acid glycine in water has a value of 1.06 10-9 m2/s. In a 2.0-cm-long tube with a cross-sectional area of 1.5 10-4 m2, the mass rate of diffusion is m/t = 4.2 ...
-
The amino acid glycine can be condensed to form a polymer called polyglycine. Draw the repeating monomer unit.
-
The amino acid glycine (H2N-CH2-COOH) can participate in the following equilibria in water: (a) Use the values of Ka and Kb to estimate the equilibrium constant for the intramolecular proton transfer...
-
2. Consider two households, one with an income twice as large as the other. Both households pay the equivalent of 5% of their income to consume free (public) education to the quantity PS (where PS...
-
Your manager is quite impressed with the security considerations you explained to her thus far, however, she asks that you give further thought to additional security measures where possible and to...
-
If a prospective client hoped to accumulate $150,000 in 10 years, how much must he or she deposit today in an account that earns an annual return of 4 percent? A. -100,614.91 B. 100,614.91 C....
-
Analyze the distribution of power in band societies and explain how conflict is resolved in them.
-
CVP analysis, changing revenues and costs Sunny Spot Travel Agency specializes in flights between Toronto and Jamaica. It books passengers on Canadian Air. Sunny Spots fixed costs are $23,500 per...
-
1. Consider the following polynomial function graph of f(x) (use the letters as a guide) a) State the interval(s) over which f'(x) is positive. b) State the interval(s) over which f"(x) is negative....
-
Thress Industries just paid a dividend of $1.50 a share (i.e., D0 = $1.50). The dividend is expected to grow 5% a year for the next 3 years and then 10% a year thereafter. What is the expected...
-
Because V is a state function, ((V/T) P /P) T = ( (V/P) T /T) P . Using this relationship, show that the isothermal compressibility and isobaric expansion coefficient are related by ( /P) T = (/T) P .
-
How would you expect a park manager of a national park that adheres to natural regulation respond to a lightning-induced forest fire? To the establishment of a noxious invasive weed species?
-
How might benchmarking be used to increase productivity in the service sector?
-
Calculate cost of capital of a stock according to CAPM model if risk-free rate is 8% , market return 12% and Beta of stock 1.5.
-
Company J has returned 15% p.a. over the past five years, versus p.a. returns of 13% and 6.5% for the equity market and Treasury Bills, respectively. You run a linear regression and determine Company...
-
Aslam Enterprises is looking to evaluate two investment proposals. The following table indicates the expected cash inflows associated with these particular investment proposals. Both dw vestments...
-
What is the Capital Adequacy Ratio and why it is important for banks/financial institutions?
-
Mr Abhishek aged 30 years, living in a rented house has 4 dependents (parents, wife and his son). He is working as an engineer in a private sector company. He wants to convert his suvings into...
-
Solve the problem. An arch of a bridge has the shape of the top half of an ellipse. The arch is 40 ft wide and 12 ft high at the center. Write an equation of the complete ellipse. Find the height of...
-
Briefly discuss the implications of the financial statement presentation project for the reporting of stockholders equity.
-
Estimate the coefficients a and b in the Dieterici equation of state from the critical constants of xenon. Calculate the pressure exerted by 1.0 mol Xe when it is confined to 1.0 dm3 at 25C.
-
Given that Som = 29.79 J K-I mol-1 for bismuth at 100 K and the following tabulated heat capacities data (D.G. Archer,]. Chem. Eng. Data 40, 1015 (1995)), compute the standard molar entropy of...
-
Estimate the standard reaction Gibbs energy of N2 (g) + 3 H2 (g) t 2 NH3 (g) at (a) 500 K, (b) 1000 K from their values at 298 K.
-
Can you draw a graph to show me how I would depict a change in the Monocentric City Model due to an inflow on the shape of Northwern cities due to the mass migration of African-Americans from the...
-
The firm's production function tell you if a twofold (threefold, etc) increase in all inputs increases output by less than twofold (less than threefold, etc.). Based on this information answer the...
-
After participating in the Application Based Activity (ABA) on the case involving allegations of fraud against the owner of a dating website, I judged the case in favor of the defendant. My decision...

Study smarter with the SolutionInn App


