The Clapeyron equation does not apply to second-order phase transitions, but there are two analogous equations, the
Question:
The Clapeyron equation does not apply to second-order phase transitions, but there are two analogous equations, the Ehrenfest equations, that do. They are:
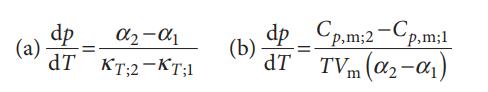
Where α is the expansion coefficient, κT the isothermal compressibility, and the subscripts 1 and 2 refer to two different phases. Derive these two equations. Why does the Clapeyron equation not apply to second-order transitions?
Transcribed Image Text:
α₂-α1 TKT;2-KT;1 dp (a) = dp_Cp,m;2-Cp,m;1 (b) dp= dT TVm (0₂-01)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
Answer The Ehrenfest equations can be derived by taking the partial derivatives of the Gibbs free en...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The law of demand does not apply to professional base-ball players. Since each team already has the maximum number of players allowed on its squad, a reduction in the wage rate that must be paid for...
-
Generally, the BFOQ defense does not apply to customer preference. But recently, some clients have been pressuring their law firms to staff their cases with female and minority lawyers. If a firm...
-
Which of the following properties does not apply to multiplication of matrices? A. Commutative B. Associative C. Distributive D. Identity
-
The Justice Department has been asked to review a merger request for a market with the following four firms. Firm Assets A .......... $156 million B .......... 130 million C .......... 45 million D...
-
ABC Toy Company earned $ 357 million of net income in 2015 and paid $ 45 million in dividends. It issued no new stock. Complete the Stockholders' Equity section for ABC Toy Company: December 31 (in...
-
Pik Corporation acquired 80 percent of Sel Corporation's common stock on January 1, 2011, for $210,000 cash. The stockholders' equity of Sel at this time consisted of $150,000 capital stock and...
-
How to use social media sites for evidence in court?
-
Rescue Sequences LLC purchased inventory' by issuing a $30,000, 10%, 60-day note on October 1. Prepare the journal entries for Rescue Sequences to record the purchase and payment assuming it uses a...
-
In general, what impacts may a firm's dividend policy, profitability, and capital intensity have on its financing requirements?
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Find the stationary distribution for the Ehrenfest Markov Chain. Data from in stationary distribution If we have an ergodic Markov Chain, we know that each state will be visited infinitely often....
-
Combine the barometric formula, p=p 0 e a/H , where H=8 km, for the dependence of the pressure on altitude, a, with the ClausiusClapeyron equation, and predict how the boiling temperature of a liquid...
-
In a laboratory experiment at 25 C, students measured the pH of samples of rainwater and household ammonia. Determine (a) [H 3 O + ] in the rainwater, with pH measured at 4.35; (b) [OH - ] in the...
-
Assume that the IT budget for a government branch office includes the following appropriation: Appropriation for: Year 1 (Current fiscal year) Year 2 Year 3 Total New software program development...
-
Write the solution using interval notation. -3z-6
-
Does Africa's colonial past affect its current economic position in the world? Why or why not? Back your points up with references only to Steger, Ellwood, Learning Materials, and resources below....
-
4. Calvin's Cleaning has employed you to investigate whether any accrual entries are needed in the business. On completion of your investigation on 30 June, you have discovered that the following...
-
Addison Company budgets production of 2,820 units during the second quarter. Information on its direct labor and its variable and fixed overhead is as follows: Direct labor Variable overhead Fixed...
-
Find an orthogonal basis for the space of the solutions to the differential equation y"' - y" + y' - y = 0 for the L2 inner product on [- , ].
-
Discuss the concept of the looking-glass self. how do you think others perceive you? do you think most people perceive you correctly?
-
A sample consisting of 1.00 mol of a van der Waals gas is compressed from 20.0 dm 3 to 10.0 dm 3 at 300 K. In the process, 20.2 kJ of work is done on the gas. Given that = {(2a/RT) b}/C p,m, with C...
-
The standard enthalpy of combustion of cyclopropane is 2091 kJ mol 1 at 25C. From this information and enthalpy of formation data for CO 2 (g) and H 2 O(g), calculate the enthalpy of formation of...
-
The standard enthalpy of formation of ethylbenzene is 12.5 kJ mol 1 . Calculate its standard enthalpy of combustion.
-
Mary was so excited about the holiday shopping season that she could hardly wait for the Black Friday sales. She got up early and spent all day at her favorite store, Mega Deals, shopping. Mary was...
-
This can be done as a group or individually. If done in a group, all group members must submit a copy of this assignment to receive a grade. If you were to open a business, what type of business...
-
6.(5pts) A total charge Q is distributed uniformly over a ring of radius R. We found an expression for the value of the electric field on the axis of the ring, E(x), a distance x from the center of...

Study smarter with the SolutionInn App


