The concentration dependence of the osmotic pressure of solutions of a macromolecule at 20C was found to
Question:
The concentration dependence of the osmotic pressure of solutions of a macromolecule at 20°C was found to be as follows:
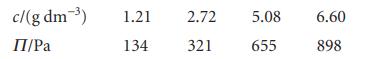
Determine the molar mass of the macromolecule and the osmotic virial coefficient.
Transcribed Image Text:
c/(g dm-3) П/Pa 1.21 134 2.72 321 5.08 655 6.60 898
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Solution P RV NB where N is the number of particles in the solution B is a consta...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A major airline manufacturer was found to be in violation of FAA safety rules and was forced to install additional safety devices in each of its planes within the next six months. The airline company...
-
A major airline manufacturer was found to be in violation of FAA safety rules and was forced to install additional safety devices in each of its planes within the next six months. The airline company...
-
A major airline manufacturer was found to be in violation of FAA safety rules and was forced to install additional safety devices in each of its planes within six months. The airline company projects...
-
Which statement does NOT reflect a way that journals require authors to disclose potential or actual conflicts of interest? Group of answer choices Require researcher's Federal tax statements Require...
-
The following hypotheses are to be tested: H0: p 0.65 HA: p > 0.65 A random sample of 500 is taken. Using each set of information following, compute the power of the test. a. = 0.01, true p = 0.68...
-
The extracts of avocado and soybean oils have been shown to slow cell inflammation in test tubes. Will taking avocado and soybean unsaponifiables (called ASU) help relieve pain for subjects with...
-
Which of the following is an extensive property? (a) Free energy (c) Specific heat (b) Refractive index (d) Surface tension.
-
Selected sales and operating data for three divisions of three different companies are given below: Required: 1. Compute the return on investment (ROI) for each division, using the formula stated in...
-
Aesthetics of Brazils, categorizing their visual arts, their music, drama ballet and other performing arts, their folklore and relevant symbols. With current references.
-
The bent rod ABDE is supported by ball-and-socket joints at A and E and by the cable DF. If a 600-N load is applied at C as shown, determine the tension in the cable. 70 mun 90 mm 600 N 110 tmm 100...
-
It is observed that the critical micelle concentration of sodium dodecyl sulfate in aqueous solution decreases as the concentration of added sodium chloride increases. Explain this effect.
-
What is the relative rate of sedimentation for two spherical particles of the same density, but which differ in radius by a factor of 10?
-
Suppose Joe, Louie, and Rebecca competes in the Bertrand ready-mix concrete market described in Section 19.2. Show that in any Nash equilibrium, all sales must occur at a price of $40 (equal to...
-
Critical ThinkingQuestions 1. Imagine that you are responsible for filling roles for a mock criminal trial. What parts would need to be represented? What types of responsibilities would each position...
-
Imagine yourself being interviewed for an entry-level police officer position by three high-ranking members of a police agency. You will respond to three different situations involving corruption....
-
A patient is referred to a consultant for diagnosis. The consultant's receptionist/secretary ob tains the patient (client) information, including the referring physician's (provider's) name. The...
-
For this discussion post, I will provide a hypothetical example that is possible in any flight school in the United States if an owner and employees are not carefully following all rules and...
-
Criminal justice professionals rely on research and crime theories to determine strategies to make improvements to policies and practices in the field. Respond to at least one of the following: Which...
-
Typical bond energies in kcal/mol are 99 for C--H, 83 for C--C, and 146 for C==C. The heat of formation of gas-phase benzene from gas-phase hydrogen and carbon atoms is -1323 kcal/mol. Calculate the...
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
A molecule in a gas undergoes about 1.0 X 109 collisions in each second. Suppose that (a) Every collision is effective in deactivating the molecule rotationally and (b) That one collision in 10 is...
-
Calculate the frequency of the J = 3 f-- 2 transition in the pure rotational spectrum of 12CI60. The equilibrium bond length is 112.81 pm.
-
If the wave number of the J = 1 f-- 0 rotational transition of IH81Br considered as a rigid rotator is 16.93 cm-1, what is (a) The moment of inertia of the molecule, (b) The bond length?
-
Subtract and simplify: 5x-6x+2-(-2-x+2x)
-
In order to value a company, you would need to forecast its future (a key word here) free cash flows. How would you approach this task? Using downloaded real data, try to make the forecast and see,...
-
share five (5) pieces of advice you would give the class about personal financial planning. Explain each. You might go back and look at the introductions in the first discussion for a general,...

Study smarter with the SolutionInn App


